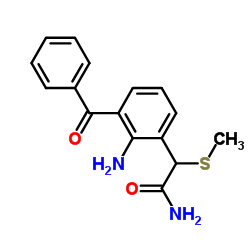
Nepafenac
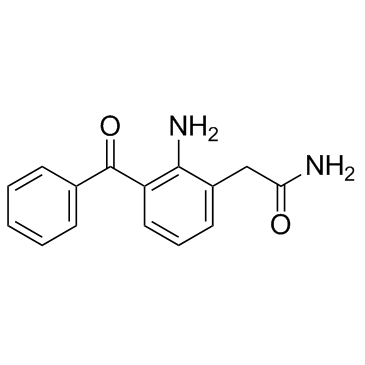
Nepafenac structure
|
Common Name | Nepafenac | ||
|---|---|---|---|---|
| CAS Number | 78281-72-8 | Molecular Weight | 254.284 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 562.5±50.0 °C at 760 mmHg | |
| Molecular Formula | C15H14N2O2 | Melting Point | 177-181ºC | |
| MSDS | USA | Flash Point | 294.0±30.1 °C | |
| Symbol |

GHS09 |
Signal Word | Warning | |
Use of NepafenacNepafenac(AHR 9434; AL 6515; Nevanac) is a selective COX-2 inhibitor; is prodrug of Amfenac.IC50 value:Target: COX-2Nepafenac is a NSAID (nonsteroidal anti inflammatory drug) that is routinely used in opthamology to control pain following cataract surgery. |
| Name | nepafenac |
|---|---|
| Synonym | More Synonyms |
| Description | Nepafenac(AHR 9434; AL 6515; Nevanac) is a selective COX-2 inhibitor; is prodrug of Amfenac.IC50 value:Target: COX-2Nepafenac is a NSAID (nonsteroidal anti inflammatory drug) that is routinely used in opthamology to control pain following cataract surgery. |
|---|---|
| Related Catalog | |
| Target |
COX-1:64.3 μM (IC50) COX-2 |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 562.5±50.0 °C at 760 mmHg |
| Melting Point | 177-181ºC |
| Molecular Formula | C15H14N2O2 |
| Molecular Weight | 254.284 |
| Flash Point | 294.0±30.1 °C |
| Exact Mass | 254.105530 |
| PSA | 86.18000 |
| LogP | 0.73 |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.641 |
| InChIKey | QEFAQIPZVLVERP-UHFFFAOYSA-N |
| SMILES | NC(=O)Cc1cccc(C(=O)c2ccccc2)c1N |
| Storage condition | Refrigerator |
|
~84% 
Nepafenac CAS#:78281-72-8 |
| Literature: Medichem, S.A. Patent: US2009/312575 A1, 2009 ; Location in patent: Page/Page column 5-6 ; |
|
~% 
Nepafenac CAS#:78281-72-8 |
| Literature: Journal of Medicinal Chemistry, , vol. 33, # 8 p. 2296 - 2304 |
|
~% 
Nepafenac CAS#:78281-72-8 |
| Literature: US5475034 A1, ; |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
| HS Code | 2924299090 |
|---|---|
| Summary | 2924299090. other cyclic amides (including cyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Use of nepafenac (Nevanac) in combination with intravitreal anti-VEGF agents in the treatment of recalcitrant exudative macular degeneration requiring monthly injections.
Clin. Ophthalmol. 4 , 1249-52, (2010) The purpose of this study is to determine the efficacy of combining topical nepafenac with monthly intravitreal injections of ranibizumab or bevacizumab in the treatment of recalcitrant exudative macu... |
| Nevanac |
| 2-(2-Amino-3-benzoylphenyl)acetamide |
| Nepafenac |
| Nepafanac |
| 2-Amino-3-benzoylbenzeneacetamide |
| 2-[2-amino-3-(phénylcarbonyl)phényl]acétamide |
| AL 6515 |
| AHR 9434 |
| 2-[2-Amino-3-(phenylcarbonyl)phenyl]acetamid |
| 2-[2-amino-3-(phenylcarbonyl)phenyl]acetamide |
| 2-Amino-3-benzoyl-phenylacetamide |