Quinacrine Dihydrochloride Dihydrate
Modify Date: 2025-08-21 15:46:46
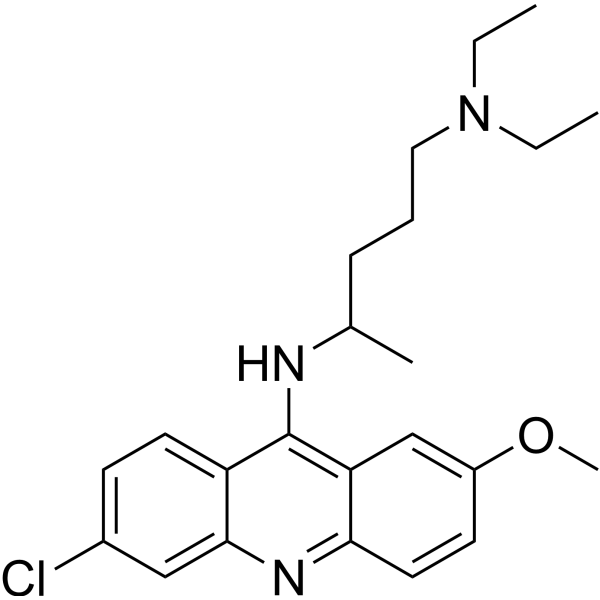
Quinacrine Dihydrochloride Dihydrate structure
|
Common Name | Quinacrine Dihydrochloride Dihydrate | ||
|---|---|---|---|---|
| CAS Number | 83-89-6 | Molecular Weight | 399.96 | |
| Density | 1.156 g/cm3 | Boiling Point | 557.1ºC at 760 mmHg | |
| Molecular Formula | C23H30ClN3O | Melting Point | 247-250ºC | |
| MSDS | N/A | Flash Point | 290.7ºC | |
Use of Quinacrine Dihydrochloride DihydrateQuinacrine (Acriquine) is an antimalarial and anti-cancer agent. Quinacrine also inhibits human aldehyde oxidase (IC50: 3.3 μM). Quinacrine has affinity for nucleic acids, and stains DNA and RNA in fixed cells (Ex/Em: 436/525 nm)[1][2][3][4][7]. |
| Name | quinacrine |
|---|---|
| Synonym | More Synonyms |
| Description | Quinacrine (Acriquine) is an antimalarial and anti-cancer agent. Quinacrine also inhibits human aldehyde oxidase (IC50: 3.3 μM). Quinacrine has affinity for nucleic acids, and stains DNA and RNA in fixed cells (Ex/Em: 436/525 nm)[1][2][3][4][7]. |
|---|---|
| Related Catalog | |
| In Vitro | Quinacrine 抑制人和兔醛氧化酶,IC50 分别为 3.3 μM 和 10 μM[2]。 Quinacrine 阻断电压依赖性钠通道 (IC50: 3.3 μM)[3]。 Quinacrine (100 μM) 也是一种 PLA2 抑制剂[4]。 Quinacrine (0-20 μM, 24 h) 抑制 SGC-7901 细胞的生长,并诱导细胞凋亡[7]。 Cell Viability AssayQuinacrine 抑制人和兔醛氧化酶,IC50 分别为 3.3 μM 和 10 μM[2]。 Cell Line: SGC-7901 cell Concentration: 0, 5, 10, 15, 20 μM Incubation Time: 24 h Result: Inhibited cell growth with an IC50 value of 16.18 μM. |
| In Vivo | Quinacrine (3-30 mg/kg,i.p.,每日一次,连续三天) 对甘油诱导的大鼠急性肾损伤具有保护作用[5]。 Quinacrine (2.5-10 mg/kg,i.p.,每天一次,连续八周) 对环孢菌素引起的大鼠肾毒性具有保护作用[6]。 Animal Model: Acute kidney injury rat model[5] Dosage: 3-30 mg/kg Administration: i.p. Result: Attenuated glycerol induced structural and functional changes in kidney. |
| References |
| Density | 1.156 g/cm3 |
|---|---|
| Boiling Point | 557.1ºC at 760 mmHg |
| Melting Point | 247-250ºC |
| Molecular Formula | C23H30ClN3O |
| Molecular Weight | 399.96 |
| Flash Point | 290.7ºC |
| Exact Mass | 399.20800 |
| PSA | 37.39000 |
| LogP | 6.04540 |
| InChIKey | GPKJTRJOBQGKQK-UHFFFAOYSA-N |
| SMILES | CCN(CC)CCCC(C)Nc1c2ccc(Cl)cc2nc2ccc(OC)cc12 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Hazard Codes | Xn: Harmful; |
|---|---|
| Risk Phrases | R36/37/38 |
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Inhibition of rabbit aldehyde oxidase
Source: ChEMBL
Target: Aldehyde oxidase 1
External Id: CHEMBL1640081
|
|
Name: NOVARTIS: Antimalarial liver stage activity measured as reduction in Plasmodium yoeli...
Source: ChEMBL
Target: Plasmodium yoelii
External Id: CHEMBL1789906
|
|
Name: NOVARTIS: Antimalarial liver stage activity measured as a greater than 50% reduction ...
Source: ChEMBL
Target: Plasmodium yoelii
External Id: CHEMBL1789905
|
|
Name: Antioxidant activity assessed as trolox equivalent of ABTS radical scavenging activit...
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL3242317
|
|
Name: Increase the activity of the Burkholderia fixLJ 2-component system
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: Burkholderia multivorans
External Id: HMS1625
|
|
Name: Cytotoxicity against human HepG2 cells assessed as cell viability after 48 hrs by ala...
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL3878513
|
|
Name: Binding affinity to Ebola virus glycoprotein assessed as dissociation constant incuba...
Source: ChEMBL
Target: N/A
External Id: CHEMBL4622624
|
|
Name: Inhibition of mouse Ido2 transfected in HEK293T cells using L-tryptophan as substrate...
Source: ChEMBL
Target: Indoleamine 2,3-dioxygenase 2
External Id: CHEMBL2210856
|
|
Name: Primary qHTS for inhibitors of NSP2Pro chikungunya virus (CHIKV)
Source: NCGC
External Id: APP-Toga-CHIKV-nsp2-p
|
Total 573, Current Page 1 of 58
1
2
3
4
5
| Quinacrine Dihydrochloride Dihydrate |
| 2-methoxy-6-chloro-9-(1-methyl-4-diethylaminobutylamino)-acridine |
| 2-methoxy-6-(trifluoromethyl)nicotinic acid |
| 2-METHOXY-6-(TRIFLUOROMETHYL)-3-PYRIDINECARBOXYLIC ACID |