2,3-Naphthalenediol

2,3-Naphthalenediol structure
|
Common Name | 2,3-Naphthalenediol | ||
|---|---|---|---|---|
| CAS Number | 92-44-4 | Molecular Weight | 160.17 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 353.9±15.0 °C at 760 mmHg | |
| Molecular Formula | C10H8O2 | Melting Point | 161-165 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 181.0±15.0 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of 2,3-NaphthalenediolNaphthalene-2,3-diol is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | naphthalene-2,3-diol |
|---|---|
| Synonym | More Synonyms |
| Description | Naphthalene-2,3-diol is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog | |
| In Vitro | 2,3-二羟基萘用于制造染料、颜料、荧光增白剂、鞣剂、抗氧化剂和防腐剂。 |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 353.9±15.0 °C at 760 mmHg |
| Melting Point | 161-165 °C(lit.) |
| Molecular Formula | C10H8O2 |
| Molecular Weight | 160.17 |
| Flash Point | 181.0±15.0 °C |
| Exact Mass | 160.052429 |
| PSA | 40.46000 |
| LogP | 2.11 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.726 |
| InChIKey | JRNGUTKWMSBIBF-UHFFFAOYSA-N |
| SMILES | Oc1cc2ccccc2cc1O |
| Storage condition | 2-8°C |
| Water Solubility | slightly soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | QJ4750000 |
| HS Code | 2907299090 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2907299090 |
|---|---|
| Summary | 2907299090 polyphenols; phenol-alcohols。supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward)。VAT:17.0%。tax rebate rate:9.0%。MFN tariff:5.5%。general tariff:30.0% |
|
Phase II metabolism in human skin: skin explants show full coverage for glucuronidation, sulfation, N-acetylation, catechol methylation, and glutathione conjugation.
Drug Metab. Dispos. 43(1) , 126-39, (2014) Although skin is the largest organ of the human body, cutaneous drug metabolism is often overlooked, and existing experimental models are insufficiently validated. This proof-of-concept study investig... |
|
|
The syntheses and characterizations of molybdenum(VI) complexes with catechol and 2,3-dihydroxynaphthalene, and the structure-effect relationship in their in vitro anticancer activities.
Dalton Trans. 41(28) , 8697-702, (2012) The reactions of (NH(4))(2)Mo(2)O(7)·2H(2)O with polyhydroxy phenols (catechol or 2,3-dihydroxynaphthalene) and ethylenediamine (en), trimethylenediamine (tn), 1,2-propanediamine (pn), triethylamine (... |
|
|
Selective antiproliferative activity of hydroxynaphthyl-beta-D-xylosides.
J. Med. Chem. 49 , 1932-8, (2006) The antiproliferative activity of the 14 isomeric monoxylosylated dihydroxynaphthalenes has been tested in vitro toward normal HFL-1 and 3T3 A31 cells as well as transformed T24 and 3T3 SV40 cells. Th... |
| 2,3-Dihydroxy naphthalene |
| 2,3-DIHYDROXY-NAPHTHALENE |
| naphthalene-2,3-diol |
| 2,3-Naphthalenediol |
| MFCD00004073 |
| EINECS 202-156-7 |
| 2,3-dihydroxynaphthalene |
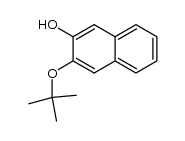 CAS#:33933-59-4
CAS#:33933-59-4![(1aS,1bR,2aS,6bR)-1a,1b,2a,6b-Tetrahydronaphtho[1,2-b:3,4-b']bisoxirene Structure](https://image.chemsrc.com/caspic/016/58692-14-1.png) CAS#:58692-14-1
CAS#:58692-14-1![Naphtho[2,3-d]-1,3-dioxole Structure](https://image.chemsrc.com/caspic/192/269-43-2.png) CAS#:269-43-2
CAS#:269-43-2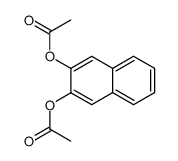 CAS#:22426-46-6
CAS#:22426-46-6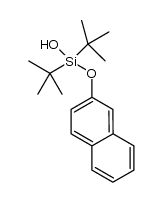 CAS#:1344113-51-4
CAS#:1344113-51-4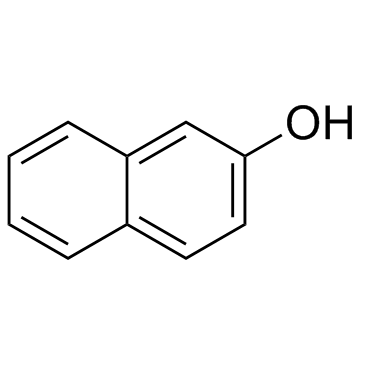 CAS#:135-19-3
CAS#:135-19-3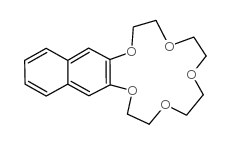 CAS#:17454-47-6
CAS#:17454-47-6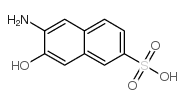 CAS#:6399-72-0
CAS#:6399-72-0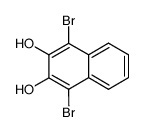 CAS#:52864-96-7
CAS#:52864-96-7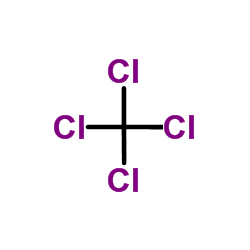 CAS#:56-23-5
CAS#:56-23-5 CAS#:142719-88-8
CAS#:142719-88-8![1H-Naphth[2,3-d]imidazole,2-methyl-(7CI,9CI) structure](https://image.chemsrc.com/caspic/089/1792-36-5.png) CAS#:1792-36-5
CAS#:1792-36-5![Benzo[b]phenazine, 5,12-dihydro structure](https://image.chemsrc.com/caspic/200/19029-32-4.png) CAS#:19029-32-4
CAS#:19029-32-4![1,4-dihydrobenzo[g]quinoxaline-2,3-dione structure](https://image.chemsrc.com/caspic/062/186666-52-4.png) CAS#:186666-52-4
CAS#:186666-52-4![12H-Benzo[b]phenoxazine structure](https://image.chemsrc.com/caspic/163/258-04-8.png) CAS#:258-04-8
CAS#:258-04-8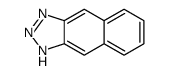 CAS#:269-12-5
CAS#:269-12-5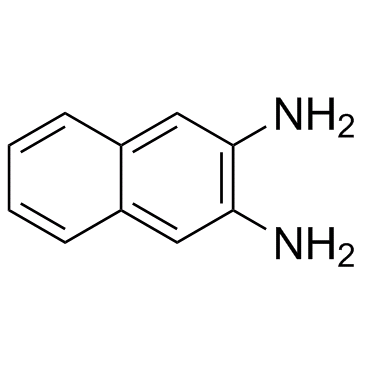 CAS#:771-97-1
CAS#:771-97-1 CAS#:5417-63-0
CAS#:5417-63-0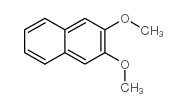 CAS#:10103-06-7
CAS#:10103-06-7
