
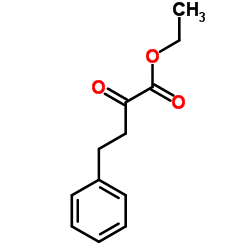
2-氧代-基丁酸乙酯结构式

|
常用名 | 2-氧代-基丁酸乙酯 | 英文名 | Ethyl 2-oxo-4-phenylbutanoate |
|---|---|---|---|---|
| CAS号 | 64920-29-2 | 分子量 | 206.238 | |
| 密度 | 1.1±0.1 g/cm3 | 沸点 | 309.0±0.0 °C at 760 mmHg | |
| 分子式 | C12H14O3 | 熔点 | N/A | |
| MSDS | 中文版 美版 | 闪点 | 140.0±19.2 °C |
|
Efficient synthesis of a chiral precursor for angiotensin-converting enzyme (ACE) inhibitors in high space-time yield by a new reductase without external cofactors.
Org. Lett. 14(8) , 1982-5, (2012) A new reductase, CgKR2, with the ability to reduce ethyl 2-oxo-4-phenylbutyrate (OPBE) to ethyl (R)-2-hydroxy-4-phenylbutyrate ((R)-HPBE), an important chiral precursor for angiotensin-converting enzyme (ACE) inhibitors, was discovered. For the first time, (R... |
|
|
Scalable biocatalytic synthesis of optically pure ethyl (R)-2-hydroxy-4-phenylbutyrate using a recombinant E. coli with high catalyst yield.
J. Biotechnol. 168(4) , 493-8, (2013) Ethyl (R)-2-hydroxy-4-phenylbutanoate [(R)-HPBE] is a versatile and important chiral intermediate for the synthesis of angiotensin-converting enzyme (ACE) inhibitors. Recombinant E. coli strain coexpressing a novel NADPH-dependent carbonyl reductase gene iolS... |
|
|
Effect of ionic liquid [BMIM][PF6] on asymmetric reduction of ethyl 2-oxo-4-phenylbutyrate by Saccharomyces cerevisiae.
J. Ind. Microbiol. Biotechnol. 35(11) , 1419-24, (2008) The effect of ionic liquid 1-butyl-3-methylimidazolium hexafluorophosphate ([BMIM][PF6]) on the asymmetric reduction of ethyl 2-oxo-4-phenylbutyrate (EOPB) to synthesize optical active ethyl 2-hydroxy-4-phenylbutyrate (EHPB) catalyzed by Saccharomyces cerevis... |
|
|
Efficient Reduction of Ethyl 2-Oxo-4-phenylbutyrate at 620 g· L- 1 by a Bacterial Reductase with Broad Substrate Spectrum. Ni Y, et al.
Adv. Synth. Catal. 353(8) , 1213-1217., (2011)
|
|
|
Efficient Reduction of Ethyl 2-Oxo-4-phenylbutyrate at 620 g· L- 1 by a Bacterial Reductase with Broad Substrate Spectrum. Ni Y, et al.
J. Mol. Catal. A: Chem. 107(1) , 85-94, (1996)
|