(-)-母菊素
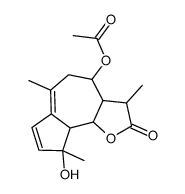
(-)-母菊素结构式

|
常用名 | (-)-母菊素 | 英文名 | matricin |
|---|---|---|---|---|
| CAS号 | 29041-35-8 | 分子量 | 306.35400 | |
| 密度 | 1.24g/cm3 | 沸点 | 481.7ºC at 760 mmHg | |
| 分子式 | C17H22O5 | 熔点 | 158-160℃ (dec.) (ethyl ether ) | |
| MSDS | 美版 | 闪点 | 175ºC |
|
Chamazulene: an antioxidant-type inhibitor of leukotriene B4 formation.
Planta Med. 60(5) , 410-3, (1994) Matricine and its transformation product chamazulene are constituents of chamomile extracts. Both have been demonstrated to exert anti-inflammatory activity in vivo. Since preparations from chamomile are used for the treatment of inflammatory skin and bowel d... |
|
|
[Pharmacologic studies on chamomile compounds. VI. Studies on the antiphlogistic effect of chamazulene and matricine].
Planta Med. 49(2) , 67-73, (1983)
|
|
|
Chamazulene carboxylic acid and matricin: a natural profen and its natural prodrug, identified through similarity to synthetic drug substances.
J. Nat. Prod. 69 , 1041-5, (2006) Chamazulene carboxylic acid (1) is a natural profen with anti-inflammatory activity and a degradation product of proazulenic sesquiterpene lactones, e.g., matricin. Both 1 and proazulenes occur in chamomile (Matricaria recutita), yarrow (Achillea millefolium)... |
|
|
Supercritical carbon dioxide extraction of chamomile flowers: extraction efficiency, stability, and in-line inclusion of chamomile-carbon dioxide extract in beta-cyclodextrin.
Phytochem. Anal. 15(4) , 249-56, (2004) The extraction of chamomile flowers using supercritical carbon dioxide was investigated with respect to extraction efficiency and compared with solvent extraction. The stability of matricine, a sensitive constituent of the essential oil of chamomile, in these... |
|
|
Absolute stereochemistry of guaianolides, of matricin and its epimers, of yarrow proazulenes, and of chamazulene carboxylic acid.
Chirality 13(7) , 337-41, (2001) Known determinations of the absolute configuration of guaianolides were collected and found to be few. The absolute configurations of guaianolides rest on the assumption that 7-H always has alpha-orientation. For matricin, only the relative configuration was ... |
|
|
On the absolute configuration of matricin.
Planta Med. 67(3) , 292-4, (2001) The assignment of the absolute configuration of matricin rests on indirect evidence. On the basis of synthetic, NMR and CD studies with its decomposition product, chamazulene carboxylic acid, we were able to firmly establish the accepted 3S,3aR,4S,9R,9aS,9bS ... |