6,7-二氢-6,6-二甲基-2-(苯基亚氨基)-1,3-苯并恶硫醇-4(5H)-酮
更新时间:2025-08-25 18:40:32
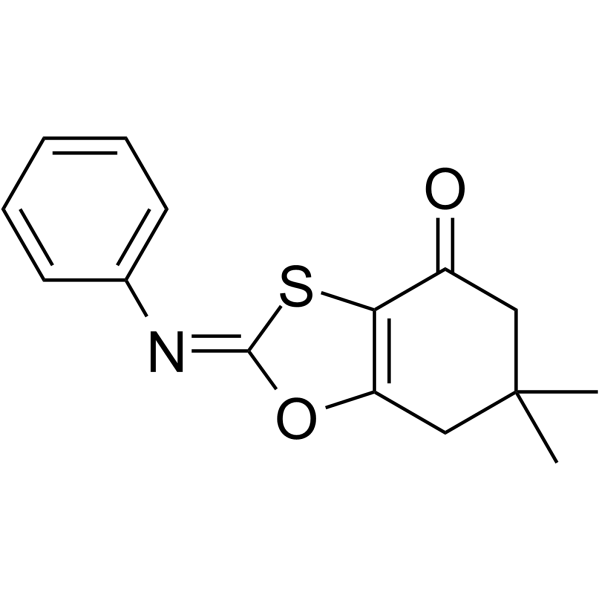
6,7-二氢-6,6-二甲基-2-(苯基亚氨基)-1,3-苯并恶硫醇-4(5H)-酮结构式

|
常用名 | 6,7-二氢-6,6-二甲基-2-(苯基亚氨基)-1,3-苯并恶硫醇-4(5H)-酮 | 英文名 | BOT-64 |
|---|---|---|---|---|
| CAS号 | 113760-29-5 | 分子量 | 273.35 | |
| 密度 | 1.27±0.1 g/cm3 | 沸点 | 402.1±48.0 °C | |
| 分子式 | C15H15NO2S | 熔点 | N/A | |
| MSDS | N/A | 闪点 | N/A |
用途BOT-64是一种抑制性κB(IκB)激酶β(IKKβ)抑制剂,IC50为1µM。BOT-64阻断脂多糖诱导的核因子-κB激活和核因子-κB调节的炎症基因转录[1]。 |
| 中文名 | NF-κB活化抑制剂VI |
|---|---|
| 英文名 | 6,6-dimethyl-2-phenylimino-5,7-dihydro-1,3-benzoxathiol-4-one |
| 英文别名 | 更多 |
| 描述 | BOT-64是一种抑制性κB(IκB)激酶β(IKKβ)抑制剂,IC50为1µM。BOT-64阻断脂多糖诱导的核因子-κB激活和核因子-κB调节的炎症基因转录[1]。 |
|---|---|
| 相关类别 | |
| 靶点实验 |
IKKβ:1 μM (IC50) |
| 体内研究 | BOT-64(3-30 mg/kg)可提高内毒素休克小鼠的存活率[1]。 |
| 参考文献 |
| 密度 | 1.27±0.1 g/cm3 |
|---|---|
| 沸点 | 402.1±48.0 °C |
| 分子式 | C15H15NO2S |
| 分子量 | 273.35 |
| 精确质量 | 273.08200 |
| PSA | 70.81000 |
| LogP | 3.72860 |
| InChIKey | XFSSJIQCIPSBHJ-UHFFFAOYSA-N |
| SMILES | CC1(C)CC(=O)c2sc(=Nc3ccccc3)oc2C1 |
| 外观性状 | 类白色固体 |
| 储存条件 | 20°C |
|
实验名称:Inhibition of NFkappaB expressed in RAW264.7 cells coexpressing NPT assessed as inhib...
来源:ChEMBL
靶标:N/A
External Id:CHEMBL2050003
|
|
实验名称:Inhibition of NFkappaB expressed in THP1 cells assessed as inhibition of LPS-induced ...
来源:ChEMBL
靶标:N/A
External Id:CHEMBL2050004
|
|
实验名称:Antiproliferative activity against human MDA-MB-231 cells by SRB assay
来源:ChEMBL
靶标:MDA-MB-231
External Id:CHEMBL2050109
|
|
实验名称:Antiproliferative activity against human HCT15 cells by SRB assay
来源:ChEMBL
靶标:HCT-15
External Id:CHEMBL2050110
|
|
实验名称:Antiproliferative activity against human ACHN cells by SRB assay
来源:ChEMBL
靶标:ACHN
External Id:CHEMBL2050005
|
|
实验名称:Antiproliferative activity against human NCI-H23 cells by SRB assay
来源:ChEMBL
靶标:NCI-H23
External Id:CHEMBL2050108
|
|
实验名称:Antiproliferative activity against human NUGC3 cells by SRB assay
来源:ChEMBL
靶标:NUGC-3
External Id:CHEMBL2050111
|
|
实验名称:Antiproliferative activity against human PC3 cells by SRB assay
来源:ChEMBL
靶标:PC-3
External Id:CHEMBL2050112
|
共8条,当前第1页,共1页
1
| 6,6-dimethyl-2-(phenylimino)-6,7-dihydro-5H-benzo[1,3]oxathiol-4-one |
| 1,3-Benzoxathiol-4(5H)-one,6,7-dihydro-6,6-dimethyl-2-(phenylimino) |
| 2-phenylimino-4-oxo-4,5,6,7-tetrahydro-6,6-dimethyl-1,3-benzoxathiole |
| 6,6-dimethyl-2-(phenylimino)-6,7-dihydrobenzo[d][1,3]oxathiol-4(5H)-one |







