1,3-Benzodioxole-5-ethanamine,hydrochloride (1:1)
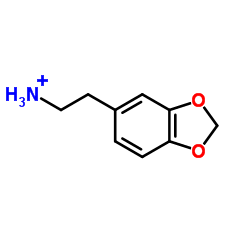
1,3-Benzodioxole-5-ethanamine,hydrochloride (1:1) structure
|
Common Name | 1,3-Benzodioxole-5-ethanamine,hydrochloride (1:1) | ||
|---|---|---|---|---|
| CAS Number | 1653-64-1 | Molecular Weight | 201.65000 | |
| Density | 1.225 | Boiling Point | 166℃ | |
| Molecular Formula | C9H12NO2 | Melting Point | 216-218 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 129.8ºC | |
|
Synthesis and antihypertensive activity of some thienylethanolamines.
J. Med. Chem. 19(7) , 876-82, (1976) Synthesis of a series of thienylethanolamines having varying substituents on the thiophene ring and on the nitrogen atom is described using the general procedure reported earlier. In the determination of their pharmacological profile, some of the derivatives ... |
|
|
An aryne route to laureline, and related topics. Gibson MS, et al.
J. Chem. Soc. Sect. C 16 , 2234-2238, (1970)
|
|
|
Determination of paramethoxyamphetamine and other amphetamine-related designer drugs by liquid chromatography/sonic spray ionization mass spectrometry.
Rapid Commun. Mass Spectrom. 16(9) , 865-70, (2002) Paramethoxyamphetamine (PMA) is an amphetamine-like designer drug that has emerged recently on the European illicit drug market. This drug has a wicked reputation, as a number of lethal intoxications have occurred. A method using high-performance liquid chrom... |
|
|
Hypothalamic-pituitary-thyroid axis and sympathetic nervous system involvement in hyperthermia induced by 3,4-methylenedioxymethamphetamine (Ecstasy).
J. Pharmacol. Exp. Ther. 305(1) , 159-66, (2003) An acute and potentially life-threatening complication associated with the recreational use of the 3,4-methylenedioxymethamphetamine (MDMA, Ecstasy) is hyperthermia. In the present study, Sprague-Dawley rats treated with MDMA (40 mg/kg s.c.) responded with a ... |
|
|
Ecstasy analogues found in cacti.
J. Psychoactive Drugs 40(2) , 219-22, (2008) Human interest in psychoactive phenethylamines is known from the use of mescaline-containing cacti and designer drugs such as Ecstasy. From the alkaloid composition of cacti we hypothesized that substances resembling Ecstasy might occur naturally. In this art... |
|
|
Oxidative stress in rat kidneys due to 3,4-methylenedioxymetamphetamine (ecstasy) toxicity.
Nephrology (Carlton.) 13(1) , 33-7, (2008) The mechanism of MDMA (3,4-methylenedioxymethamphetamine)-induced toxicity is believed to be, in part, due to enhanced oxidative stress. As MDMA is eliminated via the kidney, the aim of this study was to investigate whether MDMA created conditions of oxidativ... |
|
|
Chromatographic and spectroscopic methods of identification for the side-chain regioisomers of 3,4-methylenedioxyphenethylamines related to MDEA, MDMMA, and MBDB.
J. Chromatogr. Sci. 41(5) , 227-33, (2003) Three regioisomeric 3,4-methylenedioxyphenethylamines having the same molecular weight and major mass spectral fragments of equivalent mass have been reported as components of clandestine drug samples in recent years. These drugs of abuse are 3,4-methylenedio... |
|
|
Rat brain serotonin neurones that express neuronal nitric oxide synthase have increased sensitivity to the substituted amphetamine serotonin toxins 3,4-methylenedioxymethamphetamine and p-chloroamphetamine.
Neuroscience 134(4) , 1363-75, (2005) Substituted amphetamines such as p-chloroamphetamine and the abused drug methylenedioxymethamphetamine cause selective destruction of serotonin axons in rats, by unknown mechanisms. Since some serotonin neurones also express neuronal nitric oxide synthase, wh... |