Pyruvate kinase
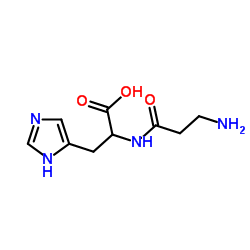
Pyruvate kinase structure
|
Common Name | Pyruvate kinase | ||
|---|---|---|---|---|
| CAS Number | 9001-59-6 | Molecular Weight | 226.232 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 656.2±55.0 °C at 760 mmHg | |
| Molecular Formula | C9H14N4O3 | Melting Point | N/A | |
| MSDS | USA | Flash Point | 350.7±31.5 °C | |
|
Quantitative phosphoproteomic analysis of T cell receptor signaling reveals system-wide modulation of protein-protein interactions.
Sci. Signal. 2 , ra46, (2009) Protein phosphorylation events during T cell receptor (TCR) signaling control the formation of complexes among proteins proximal to the TCR, the activation of kinase cascades, and the activation of transcription factors; however, the mode and extent of the in... |
|
|
Initial characterization of the human central proteome.
BMC Syst. Biol. 5 , 17, (2011) On the basis of large proteomics datasets measured from seven human cell lines we consider their intersection as an approximation of the human central proteome, which is the set of proteins ubiquitously expressed in all human cells. Composition and properties... |
|
|
Global, in vivo, and site-specific phosphorylation dynamics in signaling networks.
Cell 127(3) , 635-648, (2006) Cell signaling mechanisms often transmit information via posttranslational protein modifications, most importantly reversible protein phosphorylation. Here we develop and apply a general mass spectrometric technology for identification and quantitation of pho... |
|
|
A quantitative atlas of mitotic phosphorylation.
Proc. Natl. Acad. Sci. U. S. A. 105(31) , 10762-7, (2008) The eukaryotic cell division cycle is characterized by a sequence of orderly and highly regulated events resulting in the duplication and separation of all cellular material into two newly formed daughter cells. Protein phosphorylation by cyclin-dependent kin... |
|
|
Complete sequencing and characterization of 21,243 full-length human cDNAs.
Nat. Genet. 36 , 40-5, (2004) As a base for human transcriptome and functional genomics, we created the "full-length long Japan" (FLJ) collection of sequenced human cDNAs. We determined the entire sequence of 21,243 selected clones and found that 14,490 cDNAs (10,897 clusters) were unique... |
|
|
Immunoaffinity profiling of tyrosine phosphorylation in cancer cells.
Nat. Biotechnol. 23(1) , 94-101, (2005) Tyrosine kinases play a prominent role in human cancer, yet the oncogenic signaling pathways driving cell proliferation and survival have been difficult to identify, in part because of the complexity of the pathways and in part because of low cellular levels ... |
|
|
The status, quality, and expansion of the NIH full-length cDNA project: the Mammalian Gene Collection (MGC).
Genome Res. 14 , 2121-7, (2004) The National Institutes of Health's Mammalian Gene Collection (MGC) project was designed to generate and sequence a publicly accessible cDNA resource containing a complete open reading frame (ORF) for every human and mouse gene. The project initially used a r... |
|
|
Quantitative phosphoproteomics reveals widespread full phosphorylation site occupancy during mitosis.
Sci. Signal. 3(104) , ra3, (2010) Eukaryotic cells replicate by a complex series of evolutionarily conserved events that are tightly regulated at defined stages of the cell division cycle. Progression through this cycle involves a large number of dedicated protein complexes and signaling path... |
|
|
Lysine acetylation targets protein complexes and co-regulates major cellular functions.
Science 325(5942) , 834-40, (2009) Lysine acetylation is a reversible posttranslational modification of proteins and plays a key role in regulating gene expression. Technological limitations have so far prevented a global analysis of lysine acetylation's cellular roles. We used high-resolution... |
|
|
Lys-N and trypsin cover complementary parts of the phosphoproteome in a refined SCX-based approach.
Anal. Chem. 81(11) , 4493-501, (2009) The analysis of proteome-wide phosphorylation events is still a major analytical challenge because of the enormous complexity of protein phosphorylation networks. In this work, we evaluate the complementarity of Lys-N, Lys-C, and trypsin with regard to their ... |