h-phe-gln-val-val-cys(npys)-gly-nh2
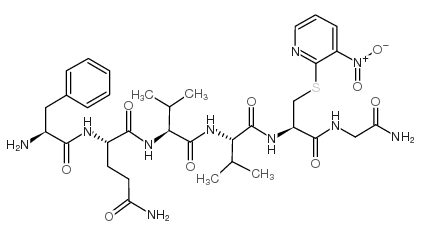
h-phe-gln-val-val-cys(npys)-gly-nh2 structure
|
Common Name | h-phe-gln-val-val-cys(npys)-gly-nh2 | ||
|---|---|---|---|---|
| CAS Number | 128102-74-9 | Molecular Weight | 772.87200 | |
| Density | 1.38g/cm3 | Boiling Point | 1152.3ºC at 760mmHg | |
| Molecular Formula | C34H48N10O9S | Melting Point | N/A | |
| MSDS | USA | Flash Point | 650.7ºC | |
|
Design and synthesis of a kininogen-based selective inhibitor of thrombin-induced platelet aggregation.
Pept. Res. 7 , 32-35, (1994) Thrombin-induced platelet aggregation has been suggested to play an important role in reocclusion following thrombolytic therapy or angioplasty for treatment of myocardial infarction. We previously demonstrated that thrombin-induced platelet aggregation is in... |
|
|
Modulation of thrombin-induced platelet aggregation by inhibition of calpain by a synthetic peptide derived from the thiol-protease inhibitory sequence of kininogens and S-(3-nitro-2-pyridinesulfenyl)-cysteine.
Eur. J. Biochem. 214 , 233-241, (1993) Thrombin-induced platelet aggregation has been suggested to play an important role in reocclusion following thrombolytic therapy of angioplasty for treatment of myocardial infarction. We previously demonstrated that aggregation of washed platelets by thrombin... |
|
|
Specificity of the sequence in Phe-Gln-Val-Val-Cys (-3-nitro-2-pyridinesulfenyl)-Gly-NH2--a selective inhibitor of thrombin-induced platelet aggregation.
Thromb. Res. 72 , 183, (1993) Thrombin-induced platelet aggregation is mediated in part by the intracellularly activated calpain expressed onto the external side of the membrane. We have previously shown that P1, Phe-Gln-Val-Val-Cys(Npys)-Gly-NH2 [Npys = 3-nitro-2-pyridinesulfenyl], an af... |