N-(4-Nitrophenyl)-L-glutamine
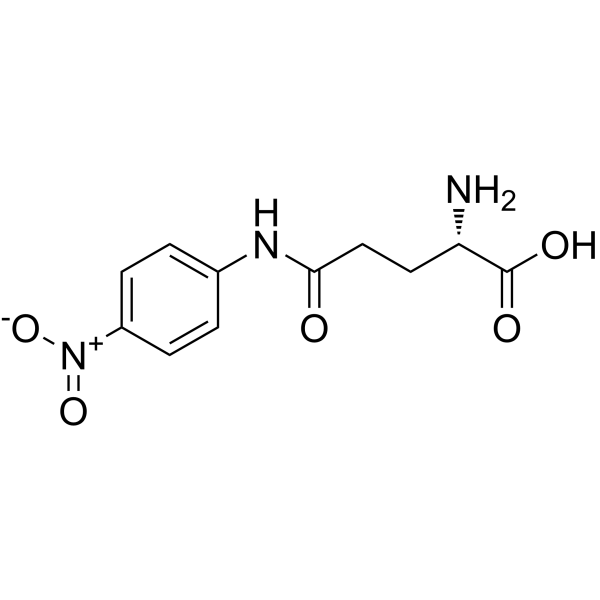
N-(4-Nitrophenyl)-L-glutamine structure
|
Common Name | N-(4-Nitrophenyl)-L-glutamine | ||
|---|---|---|---|---|
| CAS Number | 7300-59-6 | Molecular Weight | 267.238 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 590.6±50.0 °C at 760 mmHg | |
| Molecular Formula | C11H13N3O5 | Melting Point | 185-188ºC | |
| MSDS | USA | Flash Point | 311.0±30.1 °C | |
|
Donor substrate specificity of bovine kidney gamma-glutamyltransferase
Chem. Biol. Interact. 203(2) , 480-5, (2013) Highlights • Five aromatic or polycyclic aromatic glutathione conjugates were synthesized. • Enzyme kinetic studies were performed for each conjugate as a γ-glutamyltransferase (GGT) donor substrate. • All conjugates tested were substrates for GGT, with a pos... |
|
|
Rapid purification and characterization of γ-glutamyl-transpeptidase from shiitake mushroom (Lentinus edodes).
J. Food Sci. 77(6) , C640-5, (2012) γ-Glutamyl-transpeptidase (GGT) is one of the important enzymes in the pathway of odor formation in shiitake mushroom (Lentinus edodes). Rapid purification and characterization of GGT from shiitake mushroom were studied in this work. The GGT was purified 179-... |
|
|
Changes in the kinetic parameters of hepatic gamma-glutamyltransferase from streptozotocin-induced diabetic rats.
Biochim. Biophys. Acta 1545(1-2) , 184-91, (2001) Previous research has shown that the enzymatic activity of hepatic gamma-glutamyltransferase was increased in streptozotocin-induced diabetic rats with no increase in the expression of the protein. The current work has characterized the differences in the kin... |
|
|
An isopeptide bond splitting enzyme from Hirudo medicinalis similar to gamma-glutamyl transpeptidase.
Eur. J. Biochem. 256(2) , 297-302, (1998) A new enzyme from Hirudo medicinalis capable of splitting gamma-glutamyl-p-nitroanilide and Glu--Lys-(N6-gamma-glutamyllysine) (isopeptidic bond between the epsilon-amino group of lysine and the gammacarboxylic group of glutamic acid) isopeptide bonds was pur... |
|
|
Developmental changes in the activity of membrane-bound gamma-glutamyl transpeptidase and in the sialylation of synaptosomal membranes from the chick embryonic brain.
Eur. J. Biochem. 203(3) , 669-72, (1992) gamma-Glutamyl transpeptidase (GGT) is a membrane-bound sialoglycoprotein. The developmental changes in GGT activity and in sialic acid content were determined in a crude synaptosomal membrane fraction from the cerebral hemispheres of the chick embryo between... |
|
|
Role of Aggregatibacter actinomycetemcomitans in glutathione catabolism.
Oral Microbiol. Immunol. 24(3) , 236-42, (2009) Our previous studies demonstrated that three enzymes, gamma-glutamyltransferase (GGT), cysteinylglycinase (CGase) and cystalysin, are required for the catabolism of glutathione to produce hydrogen sulfide (H(2)S) in Treponema denticola. In this study, we exam... |
|
|
gamma-Glutamyltranspeptidase-catalysed acyl-transfer to the added acceptor does not proceed via the ping-pong mechanism.
Biochem. J. 304 ( Pt 3) , 869-76, (1994) Acyl-transfer catalysed by gamma-glutamyltranspeptidase from bovine kidney was studied using gamma-L- and gamma-D-Glu-p-nitroanilide as the donor and GlyGly as the acceptor. The transfer of the gamma-Glu group to GlyGly was shown to be accompanied by transfer... |
|
|
The production and characterisation of dinitrocarbanilide antibodies raised using antigen mimics.
J. Immunol. Methods 264(1-2) , 45-51, (2002) Polyclonal antibodies were produced to detect the coccidiostat nicarbazin. Due to structural constraints of the active component of nicarbazin, dinitrocarbanilide (DNC), three different compounds that shared a common substructure with DNC were used as antigen... |
|
|
Analysis of kinetic properties of gamma-glutamyl transpeptidase from rat kidney.
Gen. Physiol. Biophys. 15(5) , 403-13, (1996) The initial rate kinetics of rat kidney gamma-glutamyl transpeptidase were measured using L-gamma-glutamyl-p-nitroanilide and glycyl-glycine as the donor and the acceptor substrate, respectively. Experimental data were fitted with the initial rate equation, a... |
|
|
The reaction of oxidized and reduced glutathione (with some amino-acids) and L-gamma-glutamyl p-nitroanilide with sheep kidney tissue homogenate. An examination of the reaction products by paper chromatography.
Biochem. Soc. Trans. 19(1) , 50S, (1991)
|