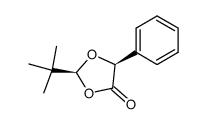
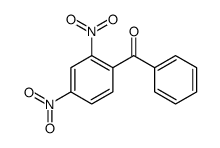
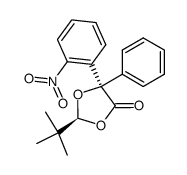
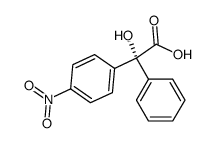
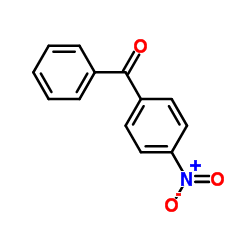
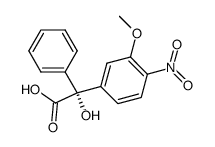
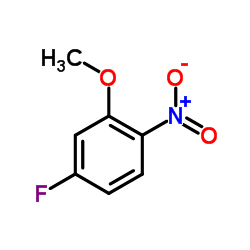
|
~% |
|
~% |
|
~89% |
|
~% |
|
~% |
|
~84% |
|
~77% |
|
~% |
|
~% |
|
~% |