Study on adsorption of glyphosate (N-phosphonomethyl glycine) pesticide on MgAl-layered double hydroxides in aqueous solution.
Feng Li, Yunfeng Wang, Qiaozhen Yang, David G Evans, Claude Forano, Xue Duan
Index: J. Hazard. Mater. 125(1-3) , 89-95, (2005)
Full Text: HTML
Abstract
MgAl-layered double hydroxides with different interlayer anions (nitrate, carbonate and chloride) were evaluated for their abilities to adsorb the organic pesticide glyphosate (N-phosphonomethyl glycine, Gly). The adsorption isotherms of Gly on layered double hydroxides (LDHs) nitrate were described by the Langmuir equation at lower equilibrium concentration of Gly (C(e)<1.0 mmol/L), and the Gly adsorption capacity on LDHs increased with the layer charge density, i.e. the structural Al3+/Mg2+ ratio. Gly adsorption on LDHs nitrate generally occurred through two processes, external surface adsorption and interlayer anion exchange. The adsorption amount on LDHs at C(e)=1.0 mmol/L decreased in the order of interlayer anions: Cl(-)>NO3(-)>CO3(2-).
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
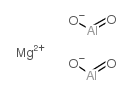 |
magnesium aluminate
CAS:12068-51-8 |
Al2H2MgO4 |
|
Epitaxially driven formation of intricate supported gold nan...
2009-12-01 [Nano Lett. 9(12) , 4258-63, (2009)] |
|
Removal of tetrafluoroborate ion from aqueous solution using...
2007-10-01 [Chemosphere 69(5) , 832-5, (2007)] |
|
Removal of HCl, SO₂, and NO by treatment of acid gas with Mg...
2011-01-01 [Chemosphere 82(4) , 587-91, (2011)] |
|
Spectrophotometric evaluation of the optical influence of co...
2009-01-01 [Dent. Mater. 25(2) , 158-65, (2009)] |
|
Removal of hydrogen chloride from gaseous streams using magn...
2008-10-01 [Chemosphere 73(5) , 844-7, (2008)] |