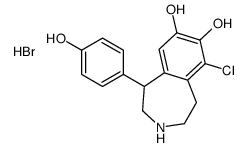
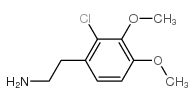
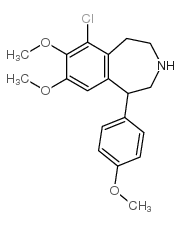
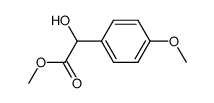
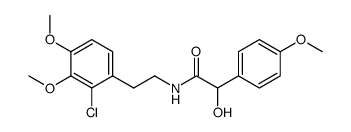
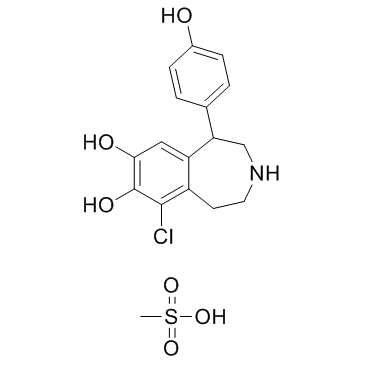
67227-56-9
| Name | fenoldopam |
|---|---|
| Synonyms |
skf 82526-j
MFCD04112986 2,3,4,5-tetrahydro-6-chloro-7,8-dihydroxy-1-(4-hydroxyphenyl)-1H-3-benzoazepine Corlopam CORLOPAM MESYLATE 6-chloro-7,8-dihydroxy-1-(p-hydroxyphenyl)-2,3,4,5-tetrahydro-1H-3-benzazepine (r)-6-chloro-2,3,4,5-tetrahydro-1-(4-hydroxyphenyl)-1h-3-benzazepine-7,8-diol 6-chloro-7,8-dihydroxy-1-(4-hydroxyphenyl)-2,3,4,5-tetrahydro-1H-3-benzazepine 6-CHLORO-2,3,4,5-TETRAHYDRO-1-(4-HYDROXYPHENYL)-1H-3-BENZAZEPINE-7,8-DIOL MESYLATE Fenoldopam EINECS 266-612-7 FENOLDOPAM HYDROCHLORIDE 6-Chloro-1-(4-hydroxyphenyl)-2,3,4,5-tetrahydro-1H-3-benzazepine-7,8-diol 6-Chloro-2,3,4,5-tetrahydro-1-(4-hydroxyphenyl)-1H-3-benzazepine-7,8-diol 6-Chloro-7,8-dihydroxy-1-p-hydroxyphenyl-2,3,4,5-tetrahydro-1H-3-benzazepine |
| Description | Fenoldopam(SKF 82526) is a drug and synthetic benzazepine derivative which acts as a selective D1 receptor partial agonist.Target: D1 ReceptorFenoldopam is a selective dopamine-1 (DA1) agonist with natriuretic/diuretic properties. Fenoldopam stimulated cAMP accumulation in LLC-PK1 cells in a dose-dependent manner, an effect which could be blocked by the DA1-selective antagonist Sch 23390. Although fenoldopam was more potent than DA (EC50 55.5 +/- 7.75 nM vs. 1.65 +/- 0.64 microM) in stimulating cAMP accumulation in LLC-PK1 cells, the maximum stimulation obtained by fenoldopam was only 37% of the maximum stimulation obtained by DA(Emax 13.0 +/- 2.95 pmol/mg of protein vs. 35.6 +/- 10.19 pmol/mg of protein) [1]. Fenoldopam is a selective dopamine1 (DA1) receptor agonist. Most of the DA1 receptor agonist activity of fenoldopam resides in the R-enantiomer, which also shows weaker alpha 2-adrenoceptor antagonist activity Fenoldopam produces vasodilation in vascular beds that are rich in vascular DA1 receptors [2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 522.6±50.0 °C at 760 mmHg |
| Molecular Formula | C16H16ClNO3 |
| Molecular Weight | 305.756 |
| Flash Point | 269.9±30.1 °C |
| Exact Mass | 305.081879 |
| PSA | 72.72000 |
| LogP | 1.72 |
| Appearance | off-white |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.656 |
| Storage condition | 2-8°C |
| Water Solubility | DMSO: >12 mg/mL |
| Hazard Codes | Xn:Harmful; |
|---|---|
| Risk Phrases | R22;R36;R42/43 |
| Safety Phrases | S22-S26-S36/37/39 |
| WGK Germany | 3 |
|
~% 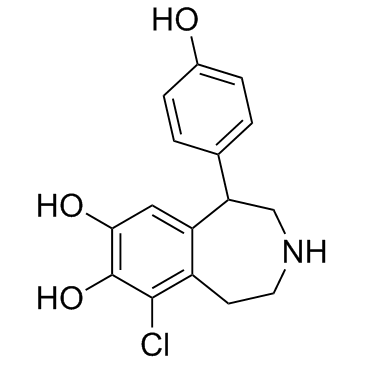
67227-56-9 |
| Literature: US2006/194967 A1, ; Page/Page column 7-8 ; |
|
~% 
67227-56-9 |
| Literature: Journal of Medicinal Chemistry, , vol. 23, # 9 p. 973 - 975 |
|
~% 
67227-56-9 |
| Literature: Journal of Medicinal Chemistry, , vol. 23, # 9 p. 973 - 975 |
|
~% 
67227-56-9 |
| Literature: Journal of Medicinal Chemistry, , vol. 23, # 9 p. 973 - 975 |
|
~% 
67227-56-9 |
| Literature: Journal of Medicinal Chemistry, , vol. 23, # 9 p. 973 - 975 |
|
~% 
67227-56-9 |
| Literature: Journal of Medicinal Chemistry, , vol. 23, # 9 p. 973 - 975 |
|
~% 
67227-56-9 |
| Literature: Journal of Medicinal Chemistry, , vol. 23, # 9 p. 973 - 975 |
|
~% 
67227-56-9 |
| Literature: Journal of Medicinal Chemistry, , vol. 23, # 9 p. 973 - 975 |
| Precursor 8 | |
|---|---|
| DownStream 1 | |