515-03-7
| Name | sclareol |
|---|---|
| Synonyms |
(1R,2R,4aS,8aS)-1-[(3R)-3-Hydroxy-3-methyl-4-penten-1-yl]-2,5,5,8a-tetramethyldecahydro-2-naphthalenol
(1R,2R,4aS,8aS)-1-[(3R)-3-Hydroxy-3-methylpent-4-en-1-yl]-2,5,5,8a-tetramethyldecahydronaphthalen-2-ol SCLAREOL(RG) EINECS 208-194-0 SCIADONIC ACID (1R,2R,8aS)-Decahydro-1-(3-hydroxy-3-methyl-4-pentenyl)-2,5,5,8a-tetramethyl-2-naphthol labd-14-ene-8,13(R)-diol MFCD00869558 SCLAREOL,NATURAL SCAREOL Sclareol (-)-sclareol SCLAREOL(P) (1R,2R,4aS,8aS)-1-[(3R)-3-Hydroxy-3-méthyl-4-pentèn-1-yl]-2,5,5,8a-tétraméthyldécahydro-2-naphtalénol |
| Description | Sclareol is isolated from Salvia sclarea with anticarcinogenic activity. Sclareol shows strong cytotoxic activity against mouse leukemia (P-388), human epidermal carcinoma (KB) cells and human leukemia cell lines. Sclareol induces cell apoptosis[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Sclareol is cytotoxic to 13 of 14 human leukemia cell lines tested, with IC50 values ranging from 6.0–24.2 μg/mL, but is not cytotoxic to peripheral blood mononuclear leukocytes within the same dose range[1]. |
| References |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 398.3±15.0 °C at 760 mmHg |
| Melting Point | 95-100 °C(lit.) |
| Molecular Formula | C20H36O2 |
| Molecular Weight | 308.499 |
| Flash Point | 169.1±15.0 °C |
| Exact Mass | 308.271515 |
| PSA | 40.46000 |
| LogP | 5.54 |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C |
| Index of Refraction | 1.490 |
| Storage condition | 2-8C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2 |
| RTECS | QK0301900 |
| HS Code | 2906199090 |
|
~0% 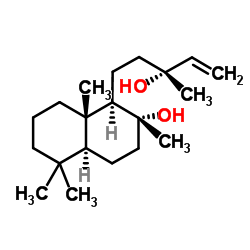
515-03-7 |
| Literature: Rogachev, Victor; Loehl, Thorsten; Markert, Thomas; Metz, Peter Arkivoc, 2012 , vol. 2012, # 3 p. 172 - 180 |
|
~% 
515-03-7 |
| Literature: Leite, Mary Ann F.; Sarragiotto, Maria Helena; Imamura, Paulo M.; Marsaioli, Anita J. Journal of Organic Chemistry, 1986 , vol. 51, # 26 p. 5409 - 5410 |
|
~% 
515-03-7 |
| Literature: Scheidegger,U. et al. Helvetica Chimica Acta, 1962 , vol. 45, p. 400 - 435 |
| Precursor 3 | |
|---|---|
| DownStream 9 | |
| HS Code | 2906199090 |
|---|---|
| Summary | 2906199090. cyclanic, cyclenic or cyclotherpenic alcohols. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:5.5%. General tariff:30.0% |


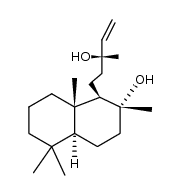



![[(1R,2R,4aS,8aS)-decahydro-2-hydroxy-2,5,5,8a-tetramethylnaphthalen-1-yl]ethanal structure](https://image.chemsrc.com/caspic/093/105014-29-7.png)

![(3R,4aS,6aR,10aR,10bS)-3-ethenyl-3,4a,7,7,10a-pentamethyl-2,5,6,6a,8,9,10,10b-octahydro-1H-benzo[f]chromene structure](https://image.chemsrc.com/caspic/327/596-84-9.png)
![(3S,6aα,10bα)-Dodecahydro-3,4aβ,7,7,10aβ-pentamethyl-3β-vinyl-1H-naphtho[2,1-b]pyran structure](https://image.chemsrc.com/caspic/450/1227-93-6.png)