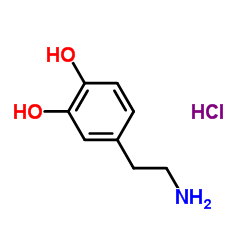
5843-65-2
| Name | (RS)-norcoclaurine |
|---|---|
| Synonyms |
Higenamine
1-(4-Hydroxybenzyl)-1,2,3,4-tetrahydroisochinolin-6,7-diol (R,S)-norcoclaurine (+-)-O-Demethylcoclaurine Coclaurine,O-demethyl-,(+-) 1-(p-hydroxybenzyl)-6,7-dihydroxy-1,2,3,4-tetrahydroisoquinoline (+-)-Demethylcoclaurine rac-norcoclaurine 1-(4-Hydroxybenzyl)-1,2,3,4-tetrahydro-6,7-isoquinolinediol (±)-HIGENAMINE Norcoclaurine 1-(4-hydroxybenzyl)-1,2,3,4-tetrahydroisoquinoline-6,7-diol 1-[(4-hydroxyphenyl)methyl]-1,2,3,4-tetrahydroisoquinoline-6,7-diol UNII:TBV5O16GAP 6,7-Isoquinolinediol,1,2,3,4-tetrahydro-1-((4-hydroxyphenyl)methyl)-,(+-) |
| Description | Higenamine (Norcoclaurine), a β2-AR agonist, is a key component of the Chinese herb aconite root that prescribes for treating symptoms of heart failure in the oriental Asian countries. Higenamine (Norcoclaurine) has anti-apoptotic effects[1][2]. |
|---|---|
| Related Catalog | |
| Target |
β2-AR[1] |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 522.4±50.0 °C at 760 mmHg |
| Melting Point | 208-210℃ |
| Molecular Formula | C16H17NO3 |
| Molecular Weight | 271.311 |
| Flash Point | 209.6±20.7 °C |
| Exact Mass | 271.120850 |
| PSA | 72.72000 |
| LogP | 1.41 |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.666 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
~99% 
5843-65-2 |
| Literature: Pesnot, Thomas; Gershater, Markus C.; Ward, John M.; Hailes, Helen C. Advanced Synthesis and Catalysis, 2012 , vol. 354, # 16 p. 2997 - 3008 |
|
~% 
5843-65-2 |
| Literature: Advanced Synthesis and Catalysis, , vol. 354, # 16 p. 2997 - 3008 |
|
~% 
5843-65-2 |
| Literature: Advanced Synthesis and Catalysis, , vol. 354, # 16 p. 2997 - 3008 |
|
~% 
5843-65-2 |
| Literature: Advanced Synthesis and Catalysis, , vol. 354, # 16 p. 2997 - 3008 |
|
~% 
5843-65-2 |
| Literature: Advanced Synthesis and Catalysis, , vol. 354, # 16 p. 2997 - 3008 |
|
~% 
5843-65-2 |
| Literature: Advanced Synthesis and Catalysis, , vol. 354, # 16 p. 2997 - 3008 |
| Precursor 7 | |
|---|---|
| DownStream 0 | |