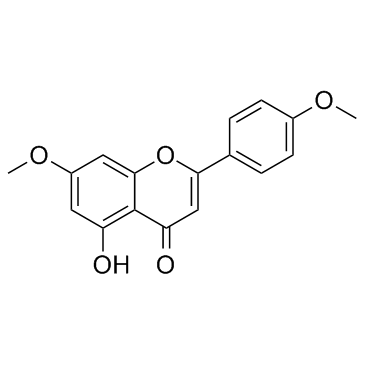
480-36-4
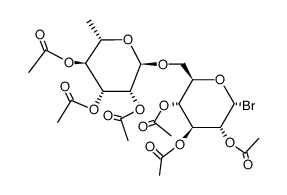
| Name | 5-hydroxy-2-(4-methoxyphenyl)-7-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxymethyl]oxan-2-yl]oxychromen-4-one |
|---|---|
| Synonyms |
Acaciin
5-Hydroxy-2-(4-methoxyphenyl)-7-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-({[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yl]oxy}methyl)tetrahydro-2H-pyran-2-yl]oxy}-4H-chromen-4-one Buddleoglucoside EINECS 207-547-6 Acacetin 7-rutinoside 5-Hydroxy-2-(4-methoxyphenyl)-4-oxo-4H-chromen-7-yl-6-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside Acacetin 7-O-rutinoside Acacetin 7-O-α-L-rhamnopyranosyl-(16)-β-D-glucopyranoside acacetin-7-O-ramnosyl-glucoside linarin Acacetin-7-O-β-D-rutinoside 5,7-Dihydroxy-4'-methoxyflavone-D-glucosido-L-rhamnoside Acacetin-b-rutinoside Linarigenin-glucoside 5-Hydroxy-2-(4-methoxyphenyl)-4-oxo-4H-chromen-7-yl 6-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside Acacetin-7-rutinoside Buddleoflavonoloside Linaric acid 7-[[6-O-(6-Deoxy-a-L-mannopyranosyl)-b-D-glucopyranosyl]oxy]-5-hydroxy-2-(4-methoxyphenyl)-4H-benzopyran-4-one Acacetin-7-O-rutinoside Buddleoside apigenin-4'-methylether-7-O-rutinoside MFCD00151178 |
| Description | Linarin (Buddleoside), isolated from the flower extract of Mentha arvensis, shows selective dose dependent inhibitory effect on acetylcholinesterase (AChE)[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 885.2±65.0 °C at 760 mmHg |
| Melting Point | 258-260ºC |
| Molecular Formula | C28H32O14 |
| Molecular Weight | 592.545 |
| Flash Point | 292.2±27.8 °C |
| Exact Mass | 592.179199 |
| PSA | 217.97000 |
| LogP | 2.10 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.694 |
| Storage condition | 2~8°C |
| Hazard Codes | Xi |
|---|
|
~54% 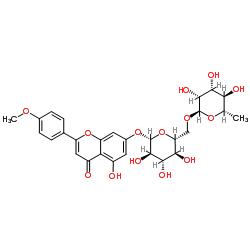
480-36-4 |
| Literature: Quintin, Jerome; Lewin, Guy Journal of Natural Products, 2004 , vol. 67, # 9 p. 1624 - 1627 |
|
~% 
480-36-4 |
| Literature: Quintin, Jerome; Lewin, Guy Journal of Natural Products, 2004 , vol. 67, # 9 p. 1624 - 1627 |
|
~% 
480-36-4 |
| Literature: Zemplen; Bognar Chemische Berichte, 1941 , vol. 74, p. 1818,1821 |