Thiophen-2-carbonitril
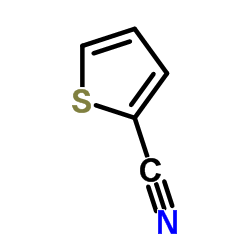
Thiophen-2-carbonitril structure
|
Common Name | Thiophen-2-carbonitril | ||
|---|---|---|---|---|
| CAS Number | 1003-31-2 | Molecular Weight | 109.149 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 193.0±13.0 °C at 760 mmHg | |
| Molecular Formula | C5H3NS | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 53.3±0.0 °C | |
| Symbol |



GHS02, GHS05, GHS07 |
Signal Word | Danger | |
| Name | 2-Thiophenecarbonitrile |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 193.0±13.0 °C at 760 mmHg |
| Molecular Formula | C5H3NS |
| Molecular Weight | 109.149 |
| Flash Point | 53.3±0.0 °C |
| Exact Mass | 108.998619 |
| PSA | 52.03000 |
| LogP | 1.27 |
| Vapour Pressure | 0.5±0.4 mmHg at 25°C |
| Index of Refraction | 1.571 |
| InChIKey | CUPOOAWTRIURFT-UHFFFAOYSA-N |
| SMILES | N#Cc1cccs1 |
| Storage condition | Flammables area |
| Symbol |



GHS02, GHS05, GHS07 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H226-H302-H315-H318-H335 |
| Precautionary Statements | P261-P280-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;Faceshields;full-face respirator (US);Gloves;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R10;R20/21/22 |
| Safety Phrases | S26-S39-S36/37-S16 |
| RIDADR | UN 1993 3/PG 3 |
| WGK Germany | 3 |
| Packaging Group | III |
| Hazard Class | 6.1 |
| HS Code | 2934999090 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Synthesis of 2,2'-bipyrroles and 2,2'-thienylpyrroles from donor-acceptor cyclopropanes and 2-cyanoheteroles.
Org. Lett. 6(6) , 1057-9, (2004) [reaction: see text] Two new series of 2,2'-bipyrroles and 2,2'-thienylpyrroles have been prepared by trimethylsilyl trifluoromethanesulfonate (TMSOTf)-mediated reaction of donor-acceptor cyclopropane... |
|
|
Understanding selectivity in the oxidative addition of the carbon-sulfur bonds of 2-cyanothiophene to Pt(0).
Inorg. Chem. 47(11) , 4596-604, (2008) The reaction of 2-cyanothiophene with a zerovalent platinum bisalkylphosphine fragment yields two thiaplatinacycles derived from the cleavage of the substituted and unsubstituted C-S bonds. While clea... |
|
|
Introduction of double amidoxime group by double post surface modification on poly(vinylbenzyl chloride) beads for higher amounts of organic dyes, As (V) and Cr (VI) removal.
J. Colloid. Interface Sci. 470 , 39-46, (2016) In this study, the synthesis of micron-sized poly(vinylbenzyl chloride) (p(VBC)) beads and subsequent conversion of the reactive chloromethyl groups to double amidoxime group containing moieties by po... |
| Thiophene-2-carbonitrile |
| 2-Thiophene acetonitrile |
| Thiophen-2-carbonitril |
| EINECS 213-706-0 |
| MFCD00005416 |
| 2-Thiophenecarbonitrile |
| 2-Cyanothiophene |
 CAS#:29683-84-9
CAS#:29683-84-9 CAS#:98-03-3
CAS#:98-03-3 CAS#:66256-03-9
CAS#:66256-03-9 CAS#:2810-04-0
CAS#:2810-04-0 CAS#:1003-09-4
CAS#:1003-09-4 CAS#:38266-87-4
CAS#:38266-87-4 CAS#:68-12-2
CAS#:68-12-2 CAS#:30717-57-8
CAS#:30717-57-8 CAS#:5813-89-8
CAS#:5813-89-8 CAS#:188290-36-0
CAS#:188290-36-0![2,5-dioctyl-3,6-di(thiophen-2-yl)pyrrolo[3,4-c] pyrrole-1,4(2H,5H)-dione structure](https://image.chemsrc.com/caspic/303/1057401-08-7.png) CAS#:1057401-08-7
CAS#:1057401-08-7 CAS#:59541-58-1
CAS#:59541-58-1 CAS#:53370-51-7
CAS#:53370-51-7 CAS#:3319-99-1
CAS#:3319-99-1 CAS#:527-72-0
CAS#:527-72-0 CAS#:492-97-7
CAS#:492-97-7 CAS#:261925-40-0
CAS#:261925-40-0 CAS#:200358-05-0
CAS#:200358-05-0 CAS#:2132-80-1
CAS#:2132-80-1
