3-Oxo-N-phenylbutanamide
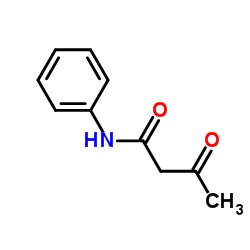
3-Oxo-N-phenylbutanamide structure
|
Common Name | 3-Oxo-N-phenylbutanamide | ||
|---|---|---|---|---|
| CAS Number | 102-01-2 | Molecular Weight | 177.200 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 329.0±44.0 °C at 760 mmHg | |
| Molecular Formula | C10H11NO2 | Melting Point | 83-88 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 152.8±28.4 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | Acetoacetanilide |
|---|---|
| Synonym | More Synonyms |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 329.0±44.0 °C at 760 mmHg |
| Melting Point | 83-88 °C(lit.) |
| Molecular Formula | C10H11NO2 |
| Molecular Weight | 177.200 |
| Flash Point | 152.8±28.4 °C |
| Exact Mass | 177.078979 |
| PSA | 46.17000 |
| LogP | 1.43 |
| Vapour density | 6.1 (vs air) |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.532 |
| InChIKey | DYRDKSSFIWVSNM-UHFFFAOYSA-N |
| SMILES | CC(=O)CC(=O)Nc1ccccc1 |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
| Water Solubility | 5 g/L (20 ºC) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H312 |
| Precautionary Statements | P280 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful |
| Risk Phrases | R21/22 |
| Safety Phrases | S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 1 |
| RTECS | AK4200000 |
| HS Code | 2924299090 |
| Precursor 6 | |
|---|---|
| DownStream 9 | |
| HS Code | 2924299090 |
|---|---|
| Summary | 2924299090. other cyclic amides (including cyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Kinetics and mechanisms of reactions between H2O2 and copper and copper oxides.
Dalton Trans. 44 , 16045-51, (2015) One of the main challenges for the nuclear power industry today is the disposal of spent nuclear fuel. One of the most developed methods for its long term storage is the Swedish KBS-3 concept where th... |
|
|
Acetoacetanilides as masked isocyanates: facile and efficient synthesis of unsymmetrically substituted ureas.
Org. Lett. 12 , 4220-4223, (2010) A general and practical method for the preparation of unsymmetrically substituted ureas has been developed. By the reactions of acetoacetanilides with various amines including primary/secondary amines... |
|
|
Massive screening yields novel and selectiveTrypanosoma cruzitriosephosphate isomerase dimer-interface-irreversible inhibitors with anti-trypanosomal activity
Eur. J. Med. Chem. 45 , 5767-72, (2010) Triosephosphate isomerase from Trypanosoma cruzi (TcTIM), an enzyme in the glycolytic pathway that exhibits high catalytic rates of glyceraldehyde-3-phosphate- and dihydroxyacetone-phosphate-isomeriza... |
| acetoacetamidobenzene |
| Linazol |
| Coupler 633 |
| Acetoacetanilide |
| a-Acetylacetanilide |
| usafek-1239 |
| 2-acetylacetanilide |
| Acetoacetanilid |
| MFCD00008780 |
| 1V1VMR |
| N-(Acetylacetyl)aniline |
| N-Phenyl-3-ketobutanamide |
| AAA |
| Cetoactanilide |
| EINECS 202-996-4 |
| (1E)-3-Oxo-N-phenylbutanimidic acid |
| b-Ketobutyranilide |
| acetoacetylaminobenzene |
| 3-Oxo-N-phenylbutanamide |
| acetoacetic anilide |
| AAN |
| acetoacetyl-phenylamine |
| Acetoacetonilide |
| 1-(N-phenylcarbamyl)propan-2-one |
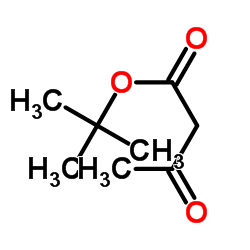 CAS#:1694-31-1
CAS#:1694-31-1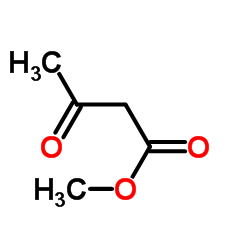 CAS#:105-45-3
CAS#:105-45-3 CAS#:5394-63-8
CAS#:5394-63-8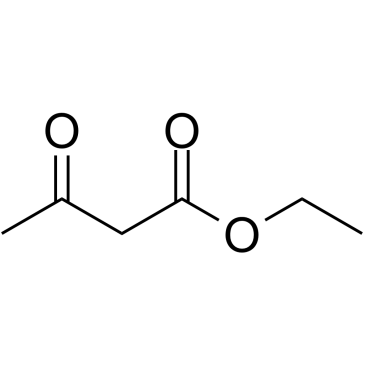 CAS#:141-97-9
CAS#:141-97-9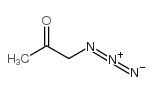 CAS#:2684-62-0
CAS#:2684-62-0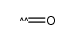 CAS#:201230-82-2
CAS#:201230-82-2 CAS#:4559-86-8
CAS#:4559-86-8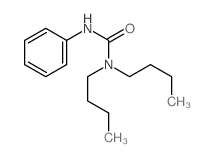 CAS#:2589-21-1
CAS#:2589-21-1 CAS#:31844-92-5
CAS#:31844-92-5 CAS#:65190-88-7
CAS#:65190-88-7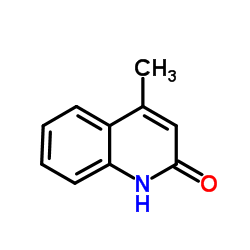 CAS#:607-66-9
CAS#:607-66-9 CAS#:141031-16-5
CAS#:141031-16-5![2-[bis(methylsulfanyl)methylidene]-3-oxo-N-phenylbutanamide structure](https://image.chemsrc.com/caspic/325/145508-18-5.png) CAS#:145508-18-5
CAS#:145508-18-5 CAS#:4344-87-0
CAS#:4344-87-0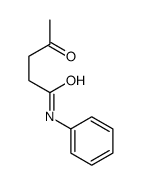 CAS#:23132-35-6
CAS#:23132-35-6
