L-penicillamine
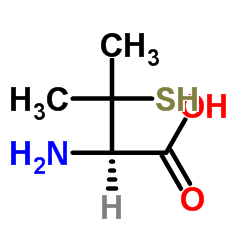
L-penicillamine structure
|
Common Name | L-penicillamine | ||
|---|---|---|---|---|
| CAS Number | 1113-41-3 | Molecular Weight | 149.211 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 251.8±35.0 °C at 760 mmHg | |
| Molecular Formula | C5H11NO2S | Melting Point | 206ºC (dec.) | |
| MSDS | Chinese USA | Flash Point | 106.1±25.9 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of L-penicillamineL-Penicillamine is a medication primarily used for the treatment of Wilson's disease. It is also used for people with kidney stones who have high urine cystine levels, rheumatoid arthritis, copper poisoning, and lead poisoning. It is taken by mouth. Common side effects include rash, loss of appetite, nausea, diarrhea, and low blood white blood cell levels. Other serious side effects include liver problems, obliterative bronchiolitis, and myasthenia gravis. It is not recommended in people with lupus erythematosus. Use during pregnancy may result in harm to the baby. Penicillamine works by binding heavy metals such that they can be removed from the body in the urine. |
| Name | L-penicillamine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 251.8±35.0 °C at 760 mmHg |
| Melting Point | 206ºC (dec.) |
| Molecular Formula | C5H11NO2S |
| Molecular Weight | 149.211 |
| Flash Point | 106.1±25.9 °C |
| Exact Mass | 149.051056 |
| PSA | 102.12000 |
| LogP | 0.93 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.528 |
| InChIKey | VVNCNSJFMMFHPL-GSVOUGTGSA-N |
| SMILES | CC(C)(S)C(N)C(=O)O |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2 |
| RTECS | YV9445500 |
| HS Code | 2930909090 |
|
~% 
L-penicillamine CAS#:1113-41-3 |
| Literature: Journal of Biological Chemistry, , vol. 184, p. 63,68 |
|
~% 
L-penicillamine CAS#:1113-41-3 |
| Literature: Journal of Biological Chemistry, , vol. 184, p. 63,68 |
|
~% 
L-penicillamine CAS#:1113-41-3 |
|
Literature: Chem.Penicillin, |
|
~% 
L-penicillamine CAS#:1113-41-3 |
| Literature: US2543358 , ; |
|
~% 
L-penicillamine CAS#:1113-41-3 |
| Literature: Journal of the Chemical Society, Faraday Transactions, , vol. 91, # 13 p. 1923 - 1930 |
|
~% 
L-penicillamine CAS#:1113-41-3 |
| Literature: Journal of the Chemical Society, Faraday Transactions, , vol. 91, # 13 p. 1923 - 1930 |
|
~% 
L-penicillamine CAS#:1113-41-3 |
| Literature: Journal of the Chemical Society, Faraday Transactions, , vol. 91, # 13 p. 1923 - 1930 |
|
~% 
L-penicillamine CAS#:1113-41-3 |
| Literature: Journal of the Chemical Society, Faraday Transactions, , vol. 91, # 13 p. 1923 - 1930 |
| Precursor 7 | |
|---|---|
| DownStream 10 | |
| HS Code | 2930909090 |
|---|---|
| Summary | 2930909090. other organo-sulphur compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Circular muscle contraction in the mice rectum plays a key role in morphine-induced constipation.
Neurogastroenterol. Motil. 26(10) , 1396-407, (2014) Although opioids induce intestinal muscle contraction and provoke constipation, the intestinal region(s) that contribute to the constipation have remained unclear. We report here a region-specific res... |
|
|
Inhibitors of bacterialN-succinyl-l,l-diaminopimelic acid desuccinylase (DapE) and demonstration of in vitro antimicrobial activity
Bioorg. Med. Chem. Lett. 19 , 6350-2, (2009) A screen biased toward compounds containing zinc-binding groups (ZBG’s) delivered a number of micromolar inhibitors of DapE from Haemophilus influenzae, including l-captopril (IC 50 = 3.3 μM, K i = 1.... |
|
|
Acetyl- and butyryl-cholinesterase inhibitory activities of the edible brown alga Eisenia bicyclis.
Arch. Pharm. Res. 38 , 1477-87, (2015) As part of our ongoing isolation of cholinesterase (ChE) inhibitors from natural marine sources, the bioactivity of the ethanolic extracts from 12 Korean seaweeds were screened for their inhibitory ac... |
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1100
|
|
Name: qHTS for Stage-Specific Inhibitors of Vaccinia Orthopoxvirus: mCherry Reporter Primar...
Source: NCGC
Target: 67.9K protein [Vaccinia virus]
External Id: Vaccinia-p2mCherry
|
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1101
|
|
Name: Cytotoxicity counterscreen for inhibitors of SARS-CoV-2 cell entry
Source: NCGC
Target: N/A
External Id: TRND-SARS-CoV-2-cytotox-48hr
|
|
Name: qHTS for Stage-Specific Inhibitors of Vaccinia Orthopoxvirus: Venus Reporter Primary ...
Source: NCGC
Target: 67.9K protein [Vaccinia virus]
External Id: Vaccinia-p2Venus
|
|
Name: Primary qHTS to identify inhibitors of SARS-CoV-2 cell entry
Source: NCGC
External Id: TRND-SARS-CoV-2-PP
|
|
Name: Primary qHTS for inhibitors of NSP2Pro chikungunya virus (CHIKV)
Source: NCGC
External Id: APP-Toga-CHIKV-nsp2-p
|
|
Name: Inhibition of Haemophilus influenzae recombinant DapE
Source: ChEMBL
Target: Succinyl-diaminopimelate desuccinylase
External Id: CHEMBL1101552
|
|
Name: Primary qHTS Assay for Inhibitors of Recombinant Selenoprotein Glutathione Peroxidase...
Source: NCGC
External Id: GPx1-biochemical-p4-p7
|
|
Name: qHTS for Inhibitors of binding or entry into cells for Marburg Virus
Source: NCGC
Target: gene 4 small orf - Marburg virus
External Id: VSVM-OFFLINE
|
| EINECS 214-203-9 |
| L(+)-Penicillamine |
| 2-Amino-3-mercapto-3-methylbutanoic acid |
| 3-mercapto-L-valine |
| 3-Sulfanyl-L-valine |
| L-β,β-Dimethylcysteine |
| L-Penicillamine |
| (R)-penicillamine |
| (±)-penicillamine |
| DL-Penicillamine |
| β, β-Dimethyl-DL-cysteine |
| β,β-Dimethyl-DL-cysteine |
| L-β-Mercaptovaline |
| (2R)-2-amino-3-mercapto-3-methyl-butyric acid |
| (2R)-2-amino-3-mercapto-3-methylbutanoic acid |
| 3,3-Dimethyl-DL-cysteine |
| 3-Sulfanylvaline |
| MFCD00064303 |
| α-AMINO-β-METHYL-β-MERCAPTOBUTYRIC ACID |
| H-Pen-OH |

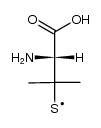



 CAS#:52-66-4
CAS#:52-66-4 CAS#:186538-00-1
CAS#:186538-00-1 CAS#:640-68-6
CAS#:640-68-6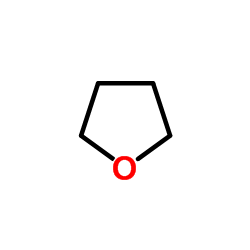 CAS#:109-99-9
CAS#:109-99-9 CAS#:72778-00-8
CAS#:72778-00-8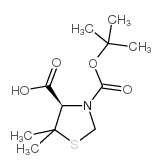 CAS#:117918-23-7
CAS#:117918-23-7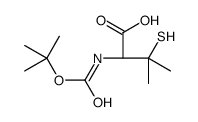 CAS#:110763-40-1
CAS#:110763-40-1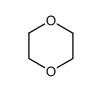 CAS#:123-91-1
CAS#:123-91-1