Azacyclonol
Modify Date: 2025-08-22 06:48:19
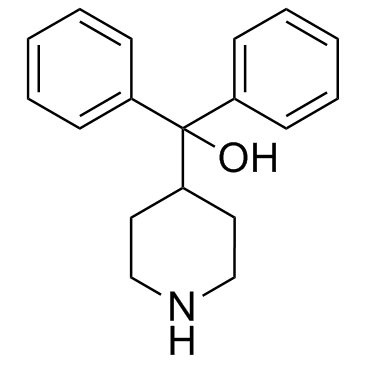
Azacyclonol structure
|
Common Name | Azacyclonol | ||
|---|---|---|---|---|
| CAS Number | 115-46-8 | Molecular Weight | 267.365 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 445.5±40.0 °C at 760 mmHg | |
| Molecular Formula | C18H21NO | Melting Point | 160-163 °C | |
| MSDS | N/A | Flash Point | 142.0±18.0 °C | |
Use of AzacyclonolAzacyclonol, also known as γ-pipradol, is a drug used to diminish hallucinations in psychotic individuals.Target: OthersAzacyclonol is a drug which is a so-called ataractive, or agent which diminishes hallucinations in psychotic individuals. The formation of Azacyclonol in human intestinal microsomes is linear with respect to time up to 60 min. The rates of formation of Azacyclonol increases linearly with microsomal protein concentration up to 2 mg/mL. The apparent Km and Vmax values of Azacyclonol are 0.82 μM and 60 pmol/min/mg protein in microsomes from human liver [1]. The formation of Azacyclonol and terfenadine alcohol from terfenadine is confirmed to be catalyzed predominantly by CYP3A(4) isozyme, and the ratio of the rate of terfenadine alcohol formation to that of Azacyclonol is 3:1 [2]. The amount of Azacyclonol eliminated renally increases on average 2-fold after rifampin dosing [3]. |
| Name | Diphenyl(piperidin-4-yl)methanol |
|---|---|
| Synonym | More Synonyms |
| Description | Azacyclonol, also known as γ-pipradol, is a drug used to diminish hallucinations in psychotic individuals.Target: OthersAzacyclonol is a drug which is a so-called ataractive, or agent which diminishes hallucinations in psychotic individuals. The formation of Azacyclonol in human intestinal microsomes is linear with respect to time up to 60 min. The rates of formation of Azacyclonol increases linearly with microsomal protein concentration up to 2 mg/mL. The apparent Km and Vmax values of Azacyclonol are 0.82 μM and 60 pmol/min/mg protein in microsomes from human liver [1]. The formation of Azacyclonol and terfenadine alcohol from terfenadine is confirmed to be catalyzed predominantly by CYP3A(4) isozyme, and the ratio of the rate of terfenadine alcohol formation to that of Azacyclonol is 3:1 [2]. The amount of Azacyclonol eliminated renally increases on average 2-fold after rifampin dosing [3]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 445.5±40.0 °C at 760 mmHg |
| Melting Point | 160-163 °C |
| Molecular Formula | C18H21NO |
| Molecular Weight | 267.365 |
| Flash Point | 142.0±18.0 °C |
| Exact Mass | 267.162323 |
| PSA | 32.26000 |
| LogP | 3.25 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.584 |
| InChIKey | ZMISODWVFHHWNR-UHFFFAOYSA-N |
| SMILES | OC(c1ccccc1)(c1ccccc1)C1CCNCC1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Hazard Codes | Xi:Irritant |
|---|---|
| Risk Phrases | R36/37/38 |
| Safety Phrases | S24/25 |
| RTECS | TN0470000 |
| HS Code | 29333999 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
|
Name: uHTS identification of cystic fibrosis induced NFkb Inhibitors in a fluoresence assay
Source: Burnham Center for Chemical Genomics
Target: cystic fibrosis transmembrane conductance regulator [Homo sapiens]
External Id: SBCCG-A764-CF-PAF-Primary-Assay
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ant...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_ANT_FLUO8_1536_1X%INH PRUN
|
|
Name: qHTS Assay for Identifying Compounds that block Entry of Ebola Virus, Screen 2 green ...
Source: NCGC
Target: N/A
External Id: Ebola screen2_EMI141217_green
|
Total 402, Current Page 1 of 41
1
2
3
4
5
| alpha,alpha-Diphenyl-4-piperidinomethanol |
| 4-(α,α Diphenyl) piperidine methanol |
| Diphenyl(4-piperidinyl)methanol |
| a,a-Diphenyl-4-piperidinemethanol |
| γ-Pipradrol |
| MFCD00066980 |
| Ataractan |
| α,α-Diphenyl-4-piperidinemethanol |
| α-(4-Piperidyl)benzhydrol |
| azacyclonol |
| diphenyl(4-piperidyl)methanol |
| Psychosan |
| g-Pipradrol |
| diphenyl(piperidin-4-yl)methanol |
| α,α-Diphenyl-4-piperidinomethanol |
| Diphenyl (g-Pyridyl)carbinol |
| a-(4-Piperidyl)benzhydrol |
| 4-(Diphenylhydroxymethyl)piperidine |
| EINECS 204-092-5 |
| Calmeran |
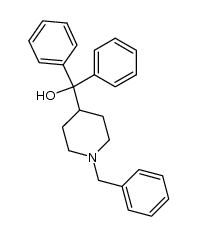 CAS#:114399-88-1
CAS#:114399-88-1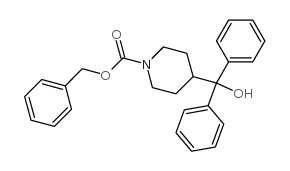 CAS#:96067-93-5
CAS#:96067-93-5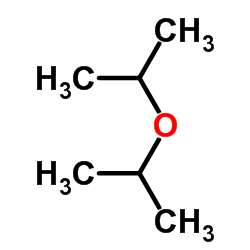 CAS#:108-20-3
CAS#:108-20-3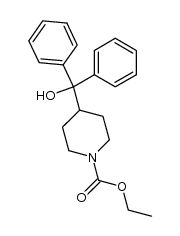 CAS#:112818-77-6
CAS#:112818-77-6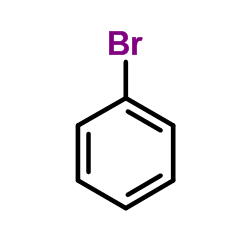 CAS#:108-86-1
CAS#:108-86-1 CAS#:24228-40-8
CAS#:24228-40-8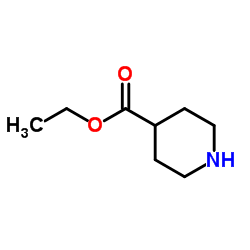 CAS#:1126-09-6
CAS#:1126-09-6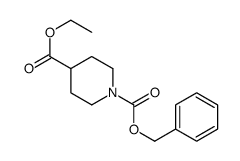 CAS#:160809-38-1
CAS#:160809-38-1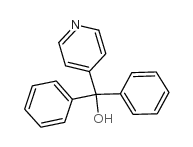 CAS#:1620-30-0
CAS#:1620-30-0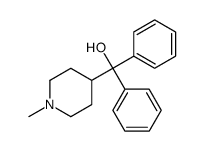 CAS#:6071-92-7
CAS#:6071-92-7![4-[4-[hydroxy(diphenyl)methyl]piperidin-1-yl]butanal structure](https://image.chemsrc.com/caspic/241/105955-84-8.png) CAS#:105955-84-8
CAS#:105955-84-8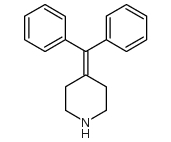 CAS#:50706-57-5
CAS#:50706-57-5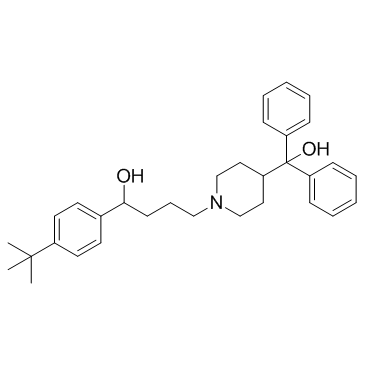 CAS#:50679-08-8
CAS#:50679-08-8 CAS#:19841-73-7
CAS#:19841-73-7 CAS#:83799-24-0
CAS#:83799-24-0 CAS#:154477-55-1
CAS#:154477-55-1 CAS#:154825-96-4
CAS#:154825-96-4![ETHYL 4-[4-[4-(HYDROXYDIPHENYLMETHYL)-1-PIPERIDINYL]-1 structure](https://image.chemsrc.com/caspic/067/169280-33-5.png) CAS#:169280-33-5
CAS#:169280-33-5![4'-tert-butyl-4-[4-(hydroxybenzhydryl)piperidino]butyrophenone structure](https://image.chemsrc.com/caspic/109/43076-30-8.png) CAS#:43076-30-8
CAS#:43076-30-8 CAS#:5704-22-3
CAS#:5704-22-3