Pheniramine maleate
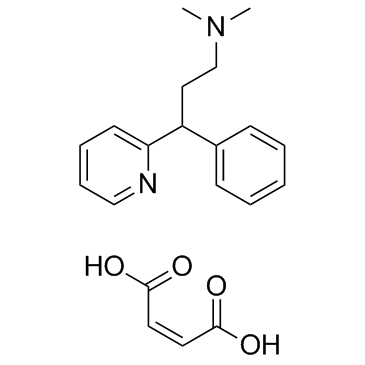
Pheniramine maleate structure
|
Common Name | Pheniramine maleate | ||
|---|---|---|---|---|
| CAS Number | 132-20-7 | Molecular Weight | 356.416 | |
| Density | N/A | Boiling Point | 348.3ºC at 760 mmHg | |
| Molecular Formula | C20H24N2O4 | Melting Point | 104-108°C | |
| MSDS | Chinese USA | Flash Point | 164.5ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Pheniramine maleatePheniramine Maleate ia an antihistamine and vasoconstrictor. |
| Name | Pheniramine Maleate |
|---|---|
| Synonym | More Synonyms |
| Description | Pheniramine Maleate ia an antihistamine and vasoconstrictor. |
|---|---|
| Related Catalog |
| Boiling Point | 348.3ºC at 760 mmHg |
|---|---|
| Melting Point | 104-108°C |
| Molecular Formula | C20H24N2O4 |
| Molecular Weight | 356.416 |
| Flash Point | 164.5ºC |
| Exact Mass | 356.173615 |
| PSA | 90.73000 |
| LogP | 2.87700 |
| Vapour Pressure | 5.07E-05mmHg at 25°C |
| InChIKey | SSOXZAQUVINQSA-BTJKTKAUSA-N |
| SMILES | CN(C)CCC(c1ccccc1)c1ccccn1.O=C(O)C=CC(=O)O |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Water Solubility | >=1 g/100 mL at 24 ºC |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Precautionary Statements | P301 + P312 + P330 |
| Hazard Codes | Xn: Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UT0175000 |
|
Assessment of protective effects of methylprednisolone and pheniramine maleate on reperfusion injury in kidney after distant organ ischemia: a rat model.
Ann. Vasc. Surg. 26(4) , 559-65, (2012) Ischemia/reperfusion (I/R) injury of tissues is a common problem that cardiovascular surgeons are faced with. Suppression of inflammation, which plays an important role in the pathogenesis of I/R inju... |
|
|
Artificial neural network combined with principal component analysis for resolution of complex pharmaceutical formulations.
Chem. Pharm. Bull. 59(1) , 35-40, (2011) A chemometric approach based on the combined use of the principal component analysis (PCA) and artificial neural network (ANN) was developed for the multicomponent determination of caffeine (CAF), mep... |
|
|
C7a, a biphosphinic cyclopalladated compound, efficiently controls the development of a patient-derived xenograft model of adult T cell leukemia/lymphoma.
Viruses 3(7) , 1041-58, (2011) Adult T-cell leukemia/lymphoma (ATLL) is a highly aggressive disease that occurs in individuals infected with the human T lymphotropic virus type 1 (HTLV-1). Patients with aggressive ATLL have a poor ... |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Inhibition of neurosphere proliferation of mouse neural precursor cells by MTT assay
Source: ChEMBL
Target: N/A
External Id: CHEMBL1266185
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Rescue cell viability in cybrid cells with a genetic mutation in complex 1 of the mit...
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: N/A
External Id: HMS1315
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Identifying Sarm1 Tir Hydrolase inhibitors through NAD-Glo assay
Source: 24386
Target: N/A
External Id: Sarm1 Tir NADase inhibitors screen
|
| (2Z)-But-2-endisäure--N,N-dimethyl-3-phenyl-3-pyridin-2-ylpropan-1-amin(1:1) |
| Pheniramine maleate |
| 1-(N,N-Dimethylamino)-3-(phenyl-3-a-pyridyl)propane Maleate |
| N,N-Dimethyl-3-phenyl-3-(pyridin-2-yl)propan-1-amine (2Z)-but-2-enedioate (1:1) |
| EINECS 205-051-4 |
| 3-Phenyl-3-(2-pyridyl)-N,N-dimethylpropylamine Maleate |
| N,N-dimethyl-3-phenyl-3-pyridin-2-ylpropan-1-amine (2Z)-but-2-enedioate |
| (Z)-but-2-enedioic acid,N,N-dimethyl-3-phenyl-3-pyridin-2-ylpropan-1-amine |
| Pheniramine maleate salt |
| 2-Pyridinepropanamine, N,N-dimethyl-γ-phenyl-, (2Z)-2-butenedioate (1:1) |
| N,N-Dimethyl-3-phenyl-3-(2-pyridinyl)-1-propanamine (2Z)-2-butenedioate (1:1) |
| acide (2Z)-but-2-ènedioïque - N,N-diméthyl-3-phényl-3-pyridin-2-ylpropan-1-amine (1:1) |
| UNII:NYW905655B |
| Naphcon A |
| MFCD00079250 |

