Phenylbutazone-13C12
Modify Date: 2025-08-27 03:46:03
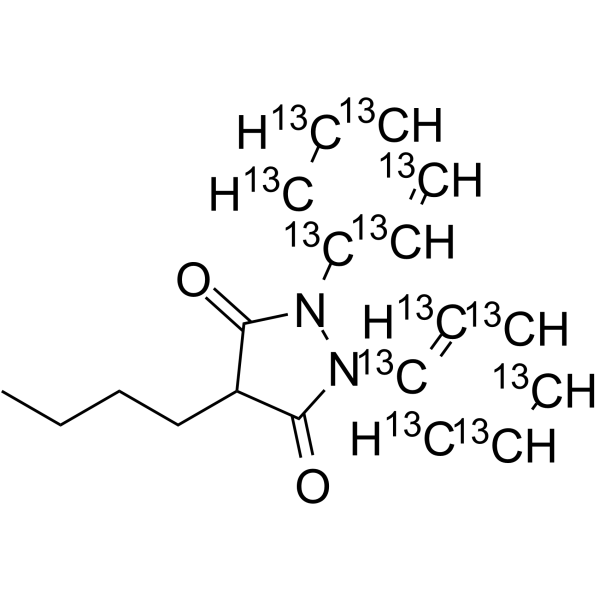
Phenylbutazone-13C12 structure
|
Common Name | Phenylbutazone-13C12 | ||
|---|---|---|---|---|
| CAS Number | 1325559-13-4 | Molecular Weight | 320.286 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C713C12H20N2O2 | Melting Point | N/A | |
| MSDS | USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Phenylbutazone-13C12Phenylbutazone-13C12 is the 13C12 labeled Phenylbutazone. Phenylbutazone is an efficient reducing cofactor for the peroxidase activity of prostaglandin H synthase (PHS). Phenylbutazone, a hepatotoxin, is a nonsteroidal anti-inflammatory drug (NSAID). Phenylbutazone induces muscle blind-like protein 1 (MBNL1) expression and has the potential for ankylosing spondylitis research. |
| Name | Phenylbutazone-13C12 |
|---|---|
| Synonym | More Synonyms |
| Description | Phenylbutazone-13C12 is the 13C12 labeled Phenylbutazone. Phenylbutazone is an efficient reducing cofactor for the peroxidase activity of prostaglandin H synthase (PHS). Phenylbutazone, a hepatotoxin, is a nonsteroidal anti-inflammatory drug (NSAID). Phenylbutazone induces muscle blind-like protein 1 (MBNL1) expression and has the potential for ankylosing spondylitis research. |
|---|---|
| Related Catalog | |
| In Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, largely as tracers for quantitation during the drug development process. Deuteration has gained attention because of its potential to affect the pharmacokinetic and metabolic profiles of drugs[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Molecular Formula | C713C12H20N2O2 |
| Molecular Weight | 320.286 |
| Exact Mass | 320.192749 |
| Index of Refraction | 1.590 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H312-H315-H319-H332-H335 |
| Precautionary Statements | P261-P280-P305 + P351 + P338 |
| Hazard Codes | Xn |
| Risk Phrases | 20/21/22-36/37/38 |
| Safety Phrases | 26-36/37 |
| RIDADR | NONH for all modes of transport |
| 4-Butyl-1,2-bis[(13C6)phenyl]-3,5-pyrazolidinedione |
| 3,5-Pyrazolidinedione, 4-butyl-1,2-di(phenyl-13C6)- |
| 4-Butyl-1,2-diphenyl-13C12-pyrazolidine-3,5-dione |
| MFCD19704819 |
| Phenylbutazone-(diphenyl-13C12) |