acenocoumarol
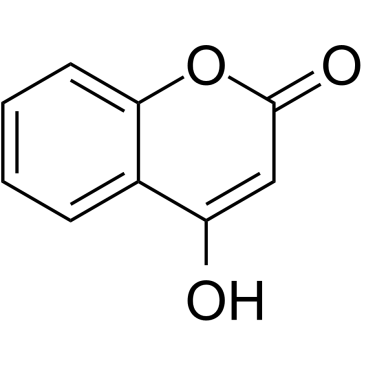
acenocoumarol structure
|
Common Name | acenocoumarol | ||
|---|---|---|---|---|
| CAS Number | 152-72-7 | Molecular Weight | 353.326 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 592.7±50.0 °C at 760 mmHg | |
| Molecular Formula | C19H15NO6 | Melting Point | 196-199ºC | |
| MSDS | Chinese USA | Flash Point | 312.3±30.1 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of acenocoumarolAcenocoumarol is an anticoagulant that functions as a Vitamin K antagonist. |
| Name | acenocoumarol |
|---|---|
| Synonym | More Synonyms |
| Description | Acenocoumarol is an anticoagulant that functions as a Vitamin K antagonist. |
|---|---|
| Related Catalog |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 592.7±50.0 °C at 760 mmHg |
| Melting Point | 196-199ºC |
| Molecular Formula | C19H15NO6 |
| Molecular Weight | 353.326 |
| Flash Point | 312.3±30.1 °C |
| Exact Mass | 353.089935 |
| PSA | 113.33000 |
| LogP | 3.15 |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.656 |
| Water Solubility | DMSO, heptane and xylene: ≥17mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319-H335 |
| Precautionary Statements | P301 + P312 + P330-P305 + P351 + P338 |
| Hazard Codes | Xn |
| Risk Phrases | 63-22-36/37/38 |
| Safety Phrases | 26-36/37 |
| RIDADR | UN 2811 |
| RTECS | GN4900000 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2932209090 |
|
~% 
acenocoumarol CAS#:152-72-7 
acenocoumarol CAS#:152-72-7 |
| Literature: Angewandte Chemie - International Edition, , vol. 42, # 40 p. 4955 - 4957 |
|
~% 
acenocoumarol CAS#:152-72-7 
acenocoumarol CAS#:152-72-7 |
| Literature: Bulletin of the Korean Chemical Society, , vol. 33, # 6 p. 1825 - 1826 |
|
~% 
acenocoumarol CAS#:152-72-7 |
| Literature: Tetrahedron Asymmetry, , vol. 12, # 5 p. 707 - 709 |
|
~% 
acenocoumarol CAS#:152-72-7 |
| Literature: Tetrahedron Asymmetry, , vol. 12, # 5 p. 707 - 709 |
| Precursor 3 | |
|---|---|
| DownStream 0 | |
| HS Code | 2932209090 |
|---|---|
| Summary | 2932209090. other lactones. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
A chiral separation strategy for acidic drugs in capillary electrochromatography using both chlorinated and nonchlorinated polysaccharide-based selectors.
Electrophoresis 35(19) , 2807-18, (2014) A generic chiral separation strategy for the analysis of acidic compounds in CEC is proposed in completion of an earlier defined strategy for nonacidic compounds. The screening step of this strategy u... |
|
|
A rapid method for determination of 22 selected drugs in human urine by UHPLC/MS/MS for clinical application.
J. AOAC Int. 97(6) , 1526-37, (2015) A rapid and sensitive ultra-HPLC/MSIMS (UHPLC/MSIMS) assay method for the simultaneous determination in human urine of 22 drugs belonging to different pharmaceutical groups was developed. The drugs we... |
|
|
Prophylaxis of experimental endocarditis with antiplatelet and antithrombin agents: a role for long-term prevention of infective endocarditis in humans?
J. Infect. Dis. 211(1) , 72-9, (2014) Infective endocarditis (IE) mostly occurs after spontaneous low-grade bacteremia. Thus, IE cannot be prevented by circumstantial antibiotic prophylaxis. Platelet activation following bacterial-fibrino... |
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
|
Name: uHTS identification of cystic fibrosis induced NFkb Inhibitors in a fluoresence assay
Source: Burnham Center for Chemical Genomics
Target: cystic fibrosis transmembrane conductance regulator [Homo sapiens]
External Id: SBCCG-A764-CF-PAF-Primary-Assay
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ant...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_ANT_FLUO8_1536_1X%INH PRUN
|
|
Name: Primary qHTS for inhibitors of NSP2Pro chikungunya virus (CHIKV)
Source: NCGC
External Id: APP-Toga-CHIKV-nsp2-p
|
| UNII:I6WP63U32H |
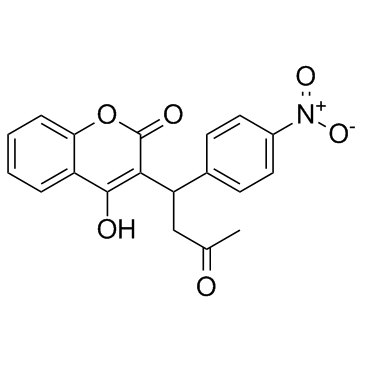
| 4-Hydroxy-3-[1-(4-nitrophenyl)-3-oxobutyl]-2H-chromen-2-one |
| Trombostop |
| EINECS 205-807-3 |
| MFCD00137816 |
| 3-(a-Acetonyl-p-nitrobenzyl)-4-hydroxycoumarin |
| 4-Hydroxy-3-[1-(4-nitrophenyl)-3-oxobutyl]-2H-1-benzopyran-2-one |
| acenocoumarol |
| 3-(a-p-Nitrophenyl-b-acetylethyl)-4-hydroxycoumarin |