U 74389G maleate
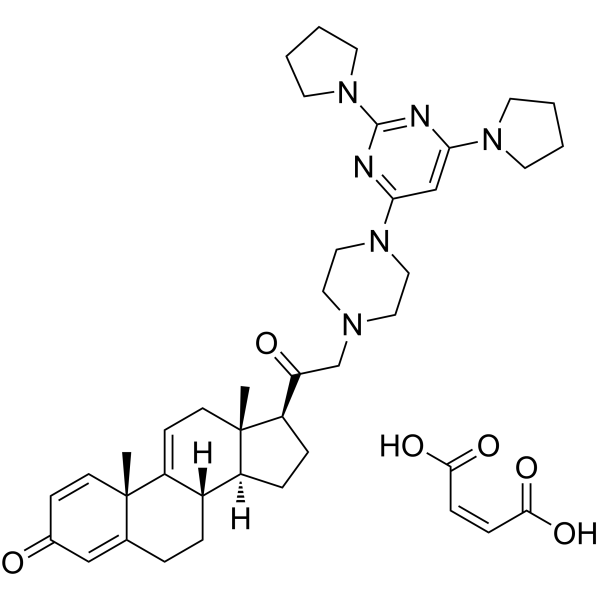
U 74389G maleate structure
|
Common Name | U 74389G maleate | ||
|---|---|---|---|---|
| CAS Number | 153190-29-5 | Molecular Weight | 726.904 | |
| Density | N/A | Boiling Point | 811.6ºC at 760 mmHg | |
| Molecular Formula | C41H54N6O6 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 444.6ºC | |
Use of U 74389G maleateU-74389G (PNU74389G meleate) is an antioxidant, can inhibit lipid peroxidation reactions. U-74389G can protect against ischemia-reperfusion injury and be widely used in animal models of ischemic injury and hypertension. U-74389G shows anti-inflammatory activity[1][2][3]. |
| Name | u-74389g |
|---|---|
| Synonym | More Synonyms |
| Description | U-74389G (PNU74389G meleate) is an antioxidant, can inhibit lipid peroxidation reactions. U-74389G can protect against ischemia-reperfusion injury and be widely used in animal models of ischemic injury and hypertension. U-74389G shows anti-inflammatory activity[1][2][3]. |
|---|---|
| Related Catalog | |
| In Vitro | U-74389G (12.5, 25 and 50 μM; 24 h) inhibits nitrite production in endotoxin stimulated peritoneal macrophages[3]. Cell Viability Assay[3] Cell Line: Peritoneal macrophages Concentration: 12.5, 25 and 50 μM Incubation Time: 24 hours Result: Reduced the nitrite concentrations in the supernatants of LPS-primed macrophages in a dose-dependent manner. |
| In Vivo | U-74389G (intravenous injection; 10 mg/kg; once daily; 6 d) treatment shows significant anti-inflammatory activity related to its ability to reduce colonic TNF-α, CMDI score, and improve weight change[2]. U-74389G (intravenous injection; 15 or 30 mg/kg) treatment significantly protects against lipopolysaccharide-induced lethality, reduces hypotension, ameliorates liver function, decreases plasma nitrite levels, restores the hyporeactivity of aortic rings to their control values and inhibits the activity of inducible NO synthase in the liver and in the aorta[3]. Animal Model: Rat model of trinitrobenzenesulfonic acid-induced colitis[2] Dosage: 10 mg/kg Administration: Intravenous injection; 10 mg/kg; once daily; 6 d Result: Reduced TNF-α, the macroscopic index of mucosal damage score (CMDI) and increased body weight. Animal Model: Male Sprague–Dawley rats injected with Lipopolysaccharide[3] Dosage: 15 or 30 mg/kg Administration: Intravenous injection; 15 or 30 mg/kg Result: Protected against lipopolysaccharide-induced lethality (90% survival rate 24 h and 80% 72 h after Lipopolysaccharide injection, respectively, following the highest dose). |
| References |
| Boiling Point | 811.6ºC at 760 mmHg |
|---|---|
| Molecular Formula | C41H54N6O6 |
| Molecular Weight | 726.904 |
| Flash Point | 444.6ºC |
| Exact Mass | 726.410461 |
| PSA | 147.48000 |
| LogP | 5.05700 |
| Vapour Pressure | 2.12E-26mmHg at 25°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
|
Contribution of nitric oxide-dependent guanylate cyclase and reactive oxygen species signaling pathways to desensitization of μ-opioid receptors in the rat locus coeruleus.
Neuropharmacology 99 , 422-31, (2015) Nitric oxide (NO) is involved in desensitization of μ-opioid receptors (MOR). We used extracellular recordings in vitro to unmask the NO-dependent pathways involved in MOR desensitization in the rat l... |
|
|
Defective mitogen-activated protein kinase (ERK2) signaling in gastric mucosa of portal hypertensive rats: potential therapeutic implications.
Hepatology 34(5) , 990-9, (2001) Portal hypertensive (PHT) gastropathy is a frequent, serious complication of liver cirrhosis. PHT gastric mucosa has numerous abnormalities such as reduced mucosal potential differences, reduced surfa... |
|
|
Copper-induced tissue factor expression in human monocytic THP-1 cells and its inhibition by antioxidants.
Circulation 92 , 238, (1995) Transition metals such as copper are known to initiate free radical formation and lipid peroxidation. Recent reports suggest that intracellular reactive oxygen intermediates can induce the transcripti... |
| MFCD01074991 |
| Pregna-1,4,9(11)-triene-3,20-dione, 21-[4-(2,6-di-1-pyrrolidinyl-4-pyrimidinyl)-1-piperazinyl]-, (2Z)-2-butenedioate (1:1) |
| 21-{4-[2,6-Di(pyrrolidin-1-yl)pyrimidin-4-yl]piperazin-1-yl}pregna-1,4,9(11)-triene-3,20-dione (2Z)-but-2-enedioate (1:1) |
| 21-{4-[2,6-Di(1-pyrrolidinyl)-4-pyrimidinyl]-1-piperazinyl}pregna-1,4,9(11)-triene-3,20-dione (2Z)-2-butenedioate (1:1) |