Ac-D-Ala-OH
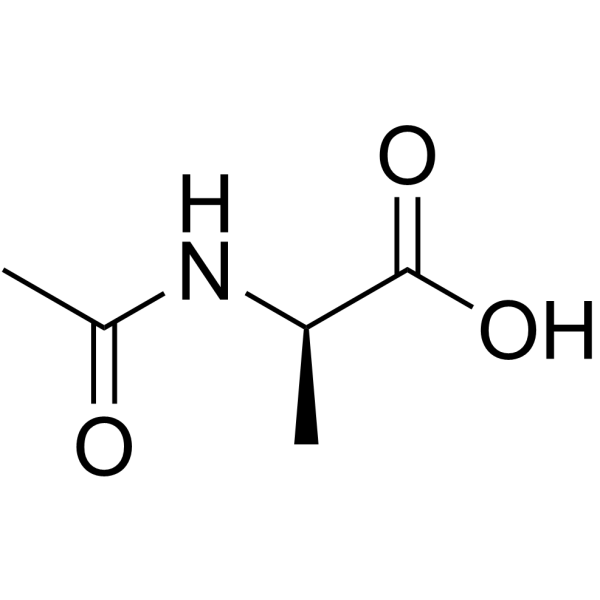
Ac-D-Ala-OH structure
|
Common Name | Ac-D-Ala-OH | ||
|---|---|---|---|---|
| CAS Number | 19436-52-3 | Molecular Weight | 131.130 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 369.7±25.0 °C at 760 mmHg | |
| Molecular Formula | C5H9NO3 | Melting Point | 128 °C | |
| MSDS | Chinese USA | Flash Point | 177.4±23.2 °C | |
Use of Ac-D-Ala-OHAc-D-Ala-OH is an alanine derivative[1]. |
| Name | (2R)-2-acetamidopropanoic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Ac-D-Ala-OH is an alanine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 369.7±25.0 °C at 760 mmHg |
| Melting Point | 128 °C |
| Molecular Formula | C5H9NO3 |
| Molecular Weight | 131.130 |
| Flash Point | 177.4±23.2 °C |
| Exact Mass | 131.058243 |
| PSA | 66.40000 |
| LogP | -1.07 |
| Appearance of Characters | Solid |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.455 |
| Storage condition | −20°C |
| Water Solubility | Slightly soluble in water. |
| Hazard Codes | Xi |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2924199090 |
| Precursor 8 | |
|---|---|
| DownStream 3 | |
| HS Code | 2924199090 |
|---|---|
| Summary | 2924199090. other acyclic amides (including acyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Substituted hippurates and hippurate analogs as substrates and inhibitors of peptidylglycine alpha-hydroxylating monooxygenase (PHM).
Bioorg. Med. Chem. 16 , 10061-74, (2008) Peptidyl alpha-hydroxylating monooxygenase (PHM) functions in vivo towards the biosynthesis of alpha-amidated peptide hormones in mammals and insects. PHM is a potential target for the development of ... |
|
|
Crystal structures of the complexes between vancomycin and cell-wall precursor analogs.
J. Mol. Biol. 385 , 1422-1432, (2009) The crystal structures of three vancomycin complexes with two vancomycin-sensitive cell-wall precursor analogs (diacetyl-Lys-D-Ala-D-Ala and acetyl-D-Ala-D-Ala) and a vancomycin-resistant cell-wall pr... |
|
|
Displacement study on a vancomycin-based stationary phase using N-acetyl-D-alanine as a competing agent.
J. Chromatogr. Sci. 40 , 83-86, (2002) The analysis of the binding data of D,L-dansyl amino acids on a vancomycin stationary phase is investigated in relation to the addition of N-acetyl-D-alanine in the mobile phase. This eluent additive ... |
| N-acetyl-D-Ala |
| AC-D-Ala-OH |
| AC-D-ALANINE |
| N-Acetyl-D-alanine-d4 |
| (2R)-2-(acetylamino)propanoic acid |
| (R)-2-(acetylamino)propanoic acid |
| (R)-2-acetamido-propanoic acid |
| N-Acetyl-D-alanine |
| Alanine, N-acetyl- |
| (R)-2-Acetamidopropanoic acid |
| acetyl-D-alanine |
| (R)-N-Acetylalanine |
| N-Acetylalanine |
| D-Alanine, N-acetyl- |
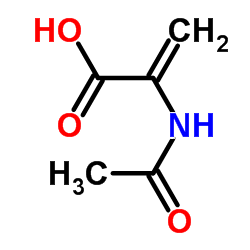 CAS#:5429-56-1
CAS#:5429-56-1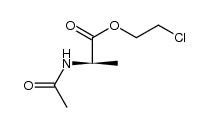 CAS#:111933-79-0
CAS#:111933-79-0 CAS#:69773-71-3
CAS#:69773-71-3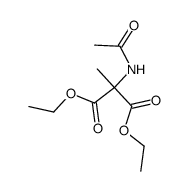 CAS#:55166-91-1
CAS#:55166-91-1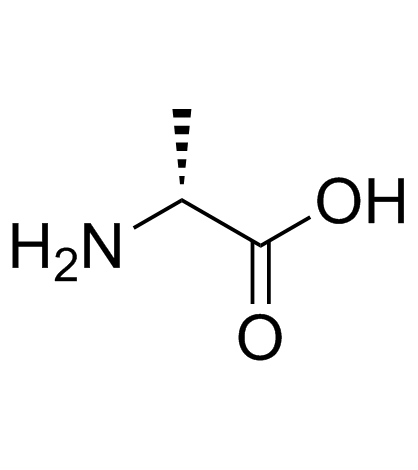 CAS#:338-69-2
CAS#:338-69-2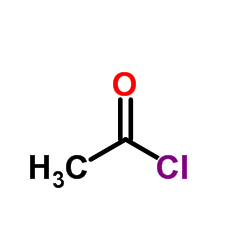 CAS#:75-36-5
CAS#:75-36-5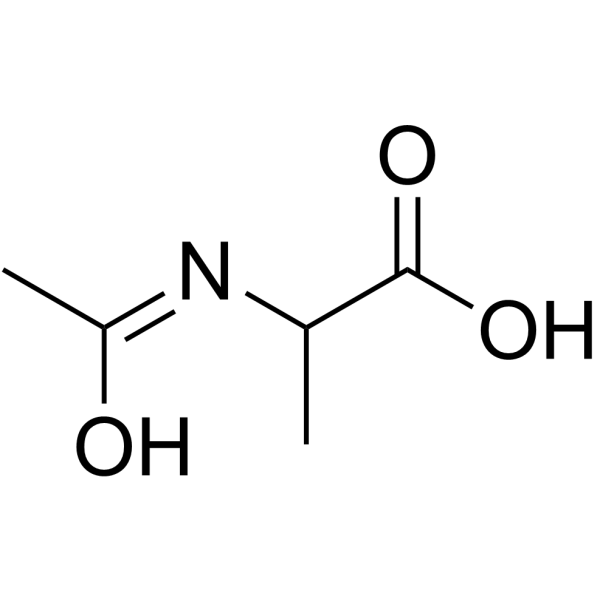 CAS#:1115-69-1
CAS#:1115-69-1 CAS#:37721-02-1
CAS#:37721-02-1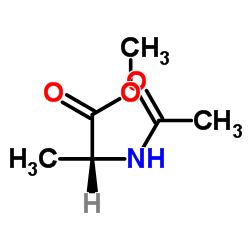 CAS#:19914-36-4
CAS#:19914-36-4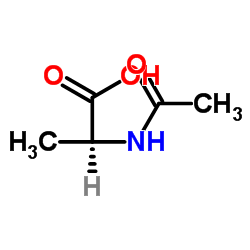 CAS#:97-69-8
CAS#:97-69-8
