Norharmane
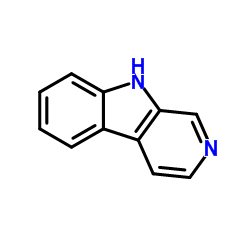
Norharmane structure
|
Common Name | Norharmane | ||
|---|---|---|---|---|
| CAS Number | 244-63-3 | Molecular Weight | 168.195 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 391.3±15.0 °C at 760 mmHg | |
| Molecular Formula | C11H8N2 | Melting Point | 219-221 ºC | |
| MSDS | Chinese USA | Flash Point | 182.1±11.7 °C | |
Use of NorharmaneNorharmane (Norharman), isolated from coffee, is a potent and selective monoamine oxidase A (MAO-A) inhibitor with a Ki of 3.34 μM[1]. |
| Name | β-carboline |
|---|---|
| Synonym | More Synonyms |
| Description | Norharmane (Norharman), isolated from coffee, is a potent and selective monoamine oxidase A (MAO-A) inhibitor with a Ki of 3.34 μM[1]. |
|---|---|
| Related Catalog | |
| Target |
MAO-A:3.34 μM (Ki) Human Endogenous Metabolite |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 391.3±15.0 °C at 760 mmHg |
| Melting Point | 219-221 ºC |
| Molecular Formula | C11H8N2 |
| Molecular Weight | 168.195 |
| Flash Point | 182.1±11.7 °C |
| Exact Mass | 168.068741 |
| PSA | 28.68000 |
| LogP | 2.80 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.785 |
| InChIKey | AIFRHYZBTHREPW-UHFFFAOYSA-N |
| SMILES | c1ccc2c(c1)[nH]c1cnccc12 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xn: Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S36/37/39-S26-S22 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UU9350000 |
| HS Code | 2933990090 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Gamma-carboline derivatives with anti-bovine viral diarrhea virus (BVDV) activity.
Bioorg. Med. Chem. 16 , 3780, (2008) Based on anti-viral screening of our heteroaromatics derived from thalidomide, the gamma-carboline skeleton has been identified as a superior scaffold structure for compounds with potent anti-bovine v... |
|
|
The use of natural product scaffolds as leads in the search for trypanothione reductase inhibitors
Bioorg. Med. Chem. 16 , 6689, (2008) Twenty-three heterocyclic compounds were evaluated for their potential as trypanothione reductase inhibitors. As a result, the harmaline, 10-thiaisoalloxazine, and aspidospermine frameworks were ident... |
|
|
Molecular insights into human monoamine oxidase (MAO) inhibition by 1,4-naphthoquinone: Evidences for menadione (vitamin K3) acting as a competitive and reversible inhibitor of MAO
Bioorg. Med. Chem. 19 , 7416-24, (2011) Monoamine oxidase (MAO) catalyzes the oxidative deamination of biogenic and exogenous amines and its inhibitors have therapeutic value for several conditions including affective disorders, stroke, neu... |
| Pyridoindole |
| CARBAZOLINE |
| b-carboline |
| β-carboline |
| MFCD00004956 |
| betacarboline |
| Norharmane |
| 9H-Pyrido[3,4-b]indole |
| EINECS 205-959-0 |
| Nor Harmane |
| norhaman |
| Norharman |
| beta-carboline |
| 9H-β-Carboline |
| 2-Azacarbazole |
 CAS#:42438-90-4
CAS#:42438-90-4 CAS#:16502-01-5
CAS#:16502-01-5![1,2,3,4-Tetrahydro-9H-pyrido[3,4-b]indol-3-carbonsaeureethylester Structure](https://image.chemsrc.com/caspic/108/84518-77-4.png) CAS#:84518-77-4
CAS#:84518-77-4 CAS#:4894-26-2
CAS#:4894-26-2 CAS#:6052-68-2
CAS#:6052-68-2 CAS#:4282-49-9
CAS#:4282-49-9![methyl 4a-(4-methyl-3,5-dioxo-1,2,4-triazolidin-1-yl)-3,4,4a,9-tetrahydro-2H-pyrido[3,4-b]indole-2-carboxylate Structure](https://image.chemsrc.com/caspic/057/557086-56-3.png) CAS#:557086-56-3
CAS#:557086-56-3 CAS#:54-12-6
CAS#:54-12-6![2-pyrido[3,4-b]indol-9-ylethanol structure](https://image.chemsrc.com/caspic/456/22946-01-6.png) CAS#:22946-01-6
CAS#:22946-01-6 CAS#:486-84-0
CAS#:486-84-0 CAS#:30684-41-4
CAS#:30684-41-4 CAS#:361202-25-7
CAS#:361202-25-7 CAS#:17337-22-3
CAS#:17337-22-3 CAS#:20127-63-3
CAS#:20127-63-3![9-(4Nitrophenyl)-9H-pyrido[3,4-b]indole structure](https://image.chemsrc.com/caspic/174/219959-85-0.png) CAS#:219959-85-0
CAS#:219959-85-0 CAS#:114719-57-2
CAS#:114719-57-2 CAS#:2521-07-5
CAS#:2521-07-5![9-Methyl-9H-Pyrido[3,4-b]indole hydrochloride structure](https://image.chemsrc.com/caspic/196/752213-27-7.png) CAS#:752213-27-7
CAS#:752213-27-7
