Dexamethasone Phosphate
Modify Date: 2025-08-26 07:10:15
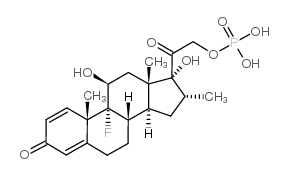
Dexamethasone Phosphate structure
|
Common Name | Dexamethasone Phosphate | ||
|---|---|---|---|---|
| CAS Number | 312-93-6 | Molecular Weight | 472.44100 | |
| Density | 1.45g/cm3 | Boiling Point | 669.6ºC at 760mmHg | |
| Molecular Formula | C22H30FO8P | Melting Point | 154-157°C (lit.) | |
| MSDS | N/A | Flash Point | 358.7ºC | |
Use of Dexamethasone PhosphateDexamethasone phosphate (Dexamethasone 21-phosphate) is a biologically inactive compound which undergoes dephosphorylation by intra-erythrocyte enzymes. The active metabolite, dexamethasone, is then released into the circulation by simple passive diffusion through cell membranes. Dexamethasone phosphate-encapsulated erythrocyte has the potential for steroid-dependent ulcerative colitis research[1]. |
| Name | dexamethasone phosphate |
|---|---|
| Synonym | More Synonyms |
| Description | Dexamethasone phosphate (Dexamethasone 21-phosphate) is a biologically inactive compound which undergoes dephosphorylation by intra-erythrocyte enzymes. The active metabolite, dexamethasone, is then released into the circulation by simple passive diffusion through cell membranes. Dexamethasone phosphate-encapsulated erythrocyte has the potential for steroid-dependent ulcerative colitis research[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.45g/cm3 |
|---|---|
| Boiling Point | 669.6ºC at 760mmHg |
| Melting Point | 154-157°C (lit.) |
| Molecular Formula | C22H30FO8P |
| Molecular Weight | 472.44100 |
| Flash Point | 358.7ºC |
| Exact Mass | 472.16600 |
| PSA | 151.17000 |
| LogP | 2.01270 |
| Index of Refraction | 1.594 |
| InChIKey | VQODGRNSFPNSQE-CXSFZGCWSA-N |
| SMILES | CC1CC2C3CCC4=CC(=O)C=CC4(C)C3(F)C(O)CC2(C)C1(O)C(=O)COP(=O)(O)O |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
|
~% 
Dexamethasone P... CAS#:312-93-6 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 22, # 7 p. 1439 - 1450 |
| Precursor 1 | |
|---|---|
| DownStream 0 | |
| HS Code | 2937290090 |
|---|
Total 31, Current Page 1 of 4
1
2
3
4
| Hexadrol phosphate |
| dexamethasone-21-phosphate |
| Dexamethasone 21-orthophosphate |
| Betnelan phosphate |
| Dexamethasone-21-dihydrogen-phosphate |
| Wymesone |
| Neodecadron |
| Oradexon phosphate |
| Dexamethasone phosphate |
| [2-[(8S,9R,10S,11S,13S,14S,16R,17R)-9-fluoro-11,17-dihydroxy-10,13,16-trimethyl-3-oxo-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-17-yl]-2-oxoethyl] dihydrogen phosphate |
