3,4-Dehydro-L-proline
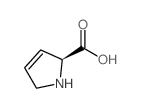
3,4-Dehydro-L-proline structure
|
Common Name | 3,4-Dehydro-L-proline | ||
|---|---|---|---|---|
| CAS Number | 4043-88-3 | Molecular Weight | 113.11 | |
| Density | 1.252g/cm3 | Boiling Point | 279.3ºC at 760mmHg | |
| Molecular Formula | C5H7NO2 | Melting Point | 239-241ºC (dec.) | |
| MSDS | Chinese USA | Flash Point | 122.7ºC | |
Use of 3,4-Dehydro-L-proline3,4-Dehydro-L-proline is aamino acids and their derivatives. |
| Name | (S)-3-Pyrroline-2-carboxylic Acid |
|---|---|
| Synonym | More Synonyms |
| Description | 3,4-Dehydro-L-proline is aamino acids and their derivatives. |
|---|---|
| Related Catalog |
| Density | 1.252g/cm3 |
|---|---|
| Boiling Point | 279.3ºC at 760mmHg |
| Melting Point | 239-241ºC (dec.) |
| Molecular Formula | C5H7NO2 |
| Molecular Weight | 113.11 |
| Flash Point | 122.7ºC |
| Exact Mass | 113.04800 |
| PSA | 49.33000 |
| Vapour Pressure | 0.00108mmHg at 25°C |
| Index of Refraction | 1.521 |
| Storage condition | -15°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UX9371345 |
| HS Code | 2933990090 |
|
~79% 
3,4-Dehydro-L-p... CAS#:4043-88-3 |
| Literature: Rueeger, Heinrich; Benn, M. H. Canadian Journal of Chemistry, 1982 , vol. 60, p. 2918 - 2920 |
|
~% 
3,4-Dehydro-L-p... CAS#:4043-88-3 |
| Literature: Rueeger, Heinrich; Benn, M. H. Canadian Journal of Chemistry, 1982 , vol. 60, p. 2918 - 2920 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Transport and signaling via the amino acid binding site of the yeast Gap1 amino acid transceptor.
Nat. Chem. Biol. 5 , 45-52, (2009) Transporter-related nutrient sensors, called transceptors, mediate nutrient activation of signaling pathways through the plasma membrane. The mechanism of action of transporting and nontransporting tr... |
|
|
Many amino acid substitutions in a hypoxia-inducible transcription factor (HIF)-1alpha-like peptide cause only minor changes in its hydroxylation by the HIF prolyl 4-hydroxylases: substitution of 3,4-dehydroproline or azetidine-2-carboxylic acid for the proline leads to a high rate of uncoupled 2-oxoglutarate decarboxylation.
J. Biol. Chem. 279(53) , 55051-9, (2004) Three human prolyl 4-hydroxylases (P4Hs) regulate the hypoxia-inducible transcription factors (HIFs) by hydroxylating a Leu-Xaa-Xaa-Leu-Ala-Pro motif. We report here that the two leucines in the Leu-G... |
|
|
Overexpression of decorin by rat arterial smooth muscle cells enhances contraction of type I collagen in vitro.
Arterioscler. Thromb. Vasc. Biol. 24(1) , 67-72, (2004) Overexpression of decorin reduces neointimal thickening in balloon-injured carotid arteries of rats by decreasing the volume of neointimal extracellular matrix (ECM). We examined the hypothesis that d... |
| 3,4-Dehydro-L-proline |
| (S)-2,5-Dihydro-1H-pyrrole-2-carboxylic acid |
| (2S)-2,5-dihydro-1H-pyrrole-2-carboxylic acid |
| EINECS 223-738-7 |
| MFCD00153487 |


 CAS#:51154-06-4
CAS#:51154-06-4 CAS#:135837-63-7
CAS#:135837-63-7
