Glycocholic acid
Modify Date: 2025-08-21 22:42:58
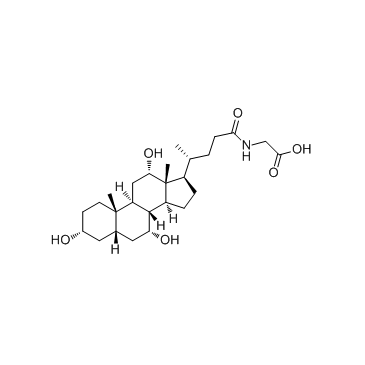
Glycocholic acid structure
|
Common Name | Glycocholic acid | ||
|---|---|---|---|---|
| CAS Number | 475-31-0 | Molecular Weight | 465.623 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 655.3±65.0 °C at 760 mmHg | |
| Molecular Formula | C26H43NO6 | Melting Point | 128°C | |
| MSDS | N/A | Flash Point | 350.1±34.3 °C | |
Use of Glycocholic acidGlycocholic acid is a bile acid with anticancer activity, targeting against pump resistance-related and non-pump resistance-related pathways. |
| Name | glycocholic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Glycocholic acid is a bile acid with anticancer activity, targeting against pump resistance-related and non-pump resistance-related pathways. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite Bax Bcl-2 MDR1 MRP1 MDR2 |
| In Vitro | Glycocholic acid (GC) increases the cytotoxicity of epirubicin, significantly increases the intracellular accumulation of epirubicin in Caco-2 cells and the absorption of epirubicin in rat small intestine, and intensified epirubicin-induced apoptosis. Glycocholic acid and epirubicin significantly reduce mRNA expression levels of human intestinal MDR1, MDR-associated protein (MRP)1, and MRP2; downregulate the MDR1 promoter region; suppress the mRNA expression of Bcl-2; induce the mRNA expression of Bax; and significantly increase the Bax-to-Bcl-2 ratio and the mRNA levels of p53, caspase-9 and -3. A combination of anticancer drugs with Glycocholic acid can control MDR via a mechanism that involves modulating P-gp and MRPs as well as regulating apoptosis-related pathways[1]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 655.3±65.0 °C at 760 mmHg |
| Melting Point | 128°C |
| Molecular Formula | C26H43NO6 |
| Molecular Weight | 465.623 |
| Flash Point | 350.1±34.3 °C |
| Exact Mass | 465.309052 |
| PSA | 127.09000 |
| LogP | 2.45 |
| Vapour Pressure | 0.0±4.5 mmHg at 25°C |
| Index of Refraction | 1.645 |
| InChIKey | RFDAIACWWDREDC-FRVQLJSFSA-N |
| SMILES | CC(CCC(=O)NCC(=O)O)C1CCC2C3C(O)CC4CC(O)CCC4(C)C3CC(O)C12C |
| Storage condition | Refrigerator |
| Water Solubility | methanol: 0.1 g/mL, clear, colorless |
| Hazard Codes | N:Dangerousfortheenvironment; |
|---|---|
| Risk Phrases | R51/53 |
| Safety Phrases | S61 |
| RIDADR | UN 3077 9/PG 3 |
| WGK Germany | 3 |
| Precursor 0 | |
|---|---|
| DownStream 3 | |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: TP_TRANSPORTER: inhibition of Ochratoxin A uptake (OTA: 1 uM, GC: 100uM) in Xenopus l...
Source: ChEMBL
Target: Solute carrier organic anion transporter family member 1A1
External Id: CHEMBL2076390
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization-based biochemical high throughput primary assay to identify...
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id: SIAE_INH_FP_1536_1X%INH PRUN
|
|
Name: p53 small molecule agonists, cell-based qHTS assay: qHTS cell viability counter scree...
Source: 824
Target: N/A
External Id: P53600
|
|
Name: p53 small molecule agonists, cell-based qHTS assay with rat liver microsomes: qHTS ce...
Source: 824
Target: N/A
External Id: P53MS958
|
|
Name: p53 small molecule agonists, cell-based qHTS assay with rat liver microsomes: Summary
Source: 824
External Id: P53MS482
|
Total 344, Current Page 1 of 35
1
2
3
4
5
| (Z)-N-[(3α,5β,7α,8ξ,12α,20R,24Z)-3,7,12,24-Tetrahydroxycholan-24-ylidene]glycine |
| N-choloyl-Glycine |
| Glycocholic Acid Hydrate |
| 3a,7a,12a-Trihydroxy-5b-cholan-24-oylglycine |
| Glycocholic acid |
| N-[(3α,5β,7α,12α)-3,7,12-Trihydroxy-24-oxocholan-24-yl]glycine |
| 2-[[(4R)-4-[(3R,5S,7R,8R,9S,10S,12S,13R,14S,17R)-3,7,12-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoyl]amino]acetic acid |
| N-Choloylglycine |
| N-[(3a,5b,7a,12a)-3,7,12-Trihydroxy-24-oxocholan-24-yl]glycine |
| EINECS 207-494-9 |
| ({(4R)-4-[(3R,5S,7R,8R,9S,10S,12S,13R,14S,17R)-3,7,12-Trihydroxy-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoyl}amino)acetic acid |
| MFCD06408004 |
 CAS#:621-79-4
CAS#:621-79-4![ethyl N-[3-phenyl-1-oxo-2-propen-1-yl] glycinate structure](https://image.chemsrc.com/caspic/018/62430-51-7.png) CAS#:62430-51-7
CAS#:62430-51-7 CAS#:56-40-6
CAS#:56-40-6
