MTOB
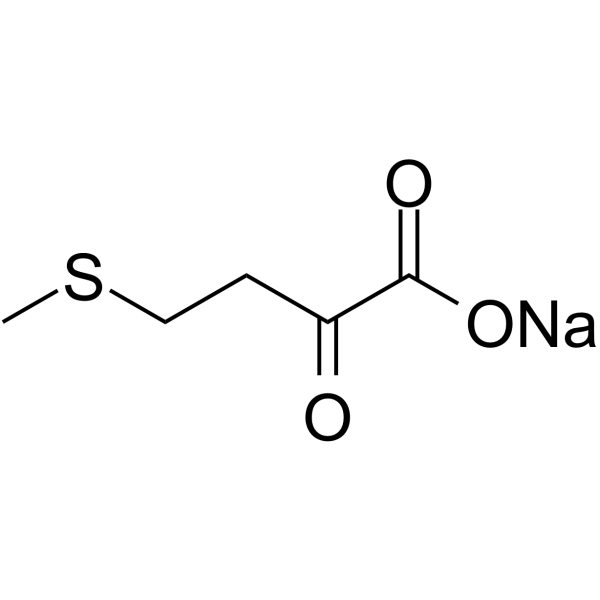
MTOB structure
|
Common Name | MTOB | ||
|---|---|---|---|---|
| CAS Number | 51828-97-8 | Molecular Weight | 170.16200 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C5H7NaO3S | Melting Point | >300ºC(lit.) | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of MTOBMTOB sodium is a potent C-terminal binding protein (CtBP) inhibitor. MTOB sodium attenuates repetitive head injury-elicited neurologic dysfunction and neuroinflammation via inhibition of the transactivation activity of CtBP1 and CtBP2. MTOB sodium antagonizes the transcriptional regulatory activity of CtBP1 and CtBP2 by eviction from their target promoters in breast cancer cell lines[1][2]. |
| Name | sodium,4-methylsulfanyl-2-oxobutanoate |
|---|---|
| Synonym | More Synonyms |
| Description | MTOB sodium is a potent C-terminal binding protein (CtBP) inhibitor. MTOB sodium attenuates repetitive head injury-elicited neurologic dysfunction and neuroinflammation via inhibition of the transactivation activity of CtBP1 and CtBP2. MTOB sodium antagonizes the transcriptional regulatory activity of CtBP1 and CtBP2 by eviction from their target promoters in breast cancer cell lines[1][2]. |
|---|---|
| Related Catalog | |
| Target |
CtBP[1] |
| In Vitro | MTOB sodium (10 mM) causes significant derepression (P<0.05) of 40% of CtBP target genes (including FGF9, CTNNB1, CEBPB, et al.) in MCF-7 and 46% in MBA-MD-231; increases the pro-epithelial E-cadherin/Vimentin ratio while reducing the pro-mesenchymal CD44/CD24 ratio, with a more significant trend (P<0.05) in MCF-7[2]. |
| In Vivo | MTOB sodium (860 mg/kg; IP, at 1 h and 18 h after the first injury) effectively suppresses the increases duration of righting reflex, and significantly decreased neurological severity score (NSS) scores[1]. Animal Model: C57BL/6 mice (traumatic brain injury)[1] Dosage: 860 mg/kg Administration: IP, at 1 h and 18 h after the first injury Result: Effectively suppressed the increased duration of righting reflex, and significantly decreased NSS scores. |
| References |
| Melting Point | >300ºC(lit.) |
|---|---|
| Molecular Formula | C5H7NaO3S |
| Molecular Weight | 170.16200 |
| Exact Mass | 170.00100 |
| PSA | 82.50000 |
| Storage condition | 2-8°C |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi |
| Risk Phrases | 36/37/38 |
| Safety Phrases | 26-36 |
| RIDADR | UN 3335 |
| HS Code | 2930909090 |
| Precursor 0 | |
|---|---|
| DownStream 3 | |
| HS Code | 2930909090 |
|---|---|
| Summary | 2930909090. other organo-sulphur compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
[Effect of substrate-dependent microbialy produced ethylene on plant growth].
Mikrobiologiia 75(2) , 277-83, (2006) Various compounds have been identified as precursors/substrates for the synthesis of ethylene (C2H4) in soil. This study was designed to compare the efficiency of four substrates, namely L-methionine ... |
|
|
Coordination of auxin and ethylene biosynthesis by the aminotransferase VAS1.
Nat. Chem. Biol. 9(4) , 244-6, (2013) We identify an Arabidopsis pyridoxal-phosphate-dependent aminotransferase, VAS1, whose loss-of-function simultaneously increases amounts of the phytohormone auxin and the ethylene precursor 1-aminocyc... |
|
|
Significant enhancement of methionol production by co-expression of the aminotransferase gene ARO8 and the decarboxylase gene ARO10 in Saccharomyces cerevisiae.
FEMS Microbiol. Lett. 362 , (2015) Methionol is an important volatile sulfur flavor compound, which can be produced via the Ehrlich pathway in Saccharomyces cerevisiae. Aminotransferase and decarboxylase are essential enzymes catalyzin... |
| KMBA |
| MFCD00010511 |
| sodium 4-methylsulfanyl-2-oxo-butyrate |
| UNII-2EZY36W75U |
| Sodium 4-(methylthio)-2-oxobutanoate |
| sodium 4-methylthio-2-oxobutyrate |
| Fema No. 3881 |
| 4-Methylthio-2-oxobutanoic acid sodium salt |
 CAS#:63-68-3
CAS#:63-68-3 CAS#:59-51-8
CAS#:59-51-8 CAS#:583-91-5
CAS#:583-91-5