Pipecolinic acid
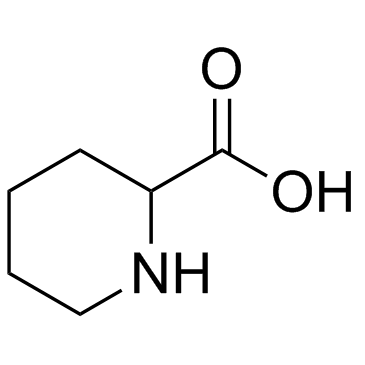
Pipecolinic acid structure
|
Common Name | Pipecolinic acid | ||
|---|---|---|---|---|
| CAS Number | 535-75-1 | Molecular Weight | 129.157 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 265.8±33.0 °C at 760 mmHg | |
| Molecular Formula | C6H11NO2 | Melting Point | 268-278ºC | |
| MSDS | Chinese USA | Flash Point | 114.5±25.4 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Pipecolinic acidPipecolic acid is a metabolite of lysine. |
| Name | DL-Pipecolinic Acid |
|---|---|
| Synonym | More Synonyms |
| Description | Pipecolic acid is a metabolite of lysine. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 265.8±33.0 °C at 760 mmHg |
| Melting Point | 268-278ºC |
| Molecular Formula | C6H11NO2 |
| Molecular Weight | 129.157 |
| Flash Point | 114.5±25.4 °C |
| Exact Mass | 129.078979 |
| PSA | 49.33000 |
| LogP | 0.00 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.479 |
| InChIKey | HXEACLLIILLPRG-UHFFFAOYSA-N |
| SMILES | O=C(O)C1CCCCN1 |
| Water Solubility | soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | 26-36-24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | TK6021000 |
| HS Code | 2933399090 |
| Precursor 0 | |
|---|---|
| DownStream 1 | |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression.
Nature 457(7231) , 910-4, (2009) Multiple, complex molecular events characterize cancer development and progression. Deciphering the molecular networks that distinguish organ-confined disease from metastatic disease may lead to the i... |
|
|
Automated screening of urine samples for carbohydrates, organic and amino acids after treatment with urease.
J. Chromatogr. A. 562(1-2) , 125-38, (1991) Eighty-five clinical urine samples and nineteen urine samples previously found by other laboratories to suggest genetic metabolic defects were prepared for trimethylsilylation by treatment with urease... |
|
|
ECNI GC-MS analysis of picolinic and quinolinic acids and their amides in human plasma, CSF, and brain tissue.
Adv. Exp. Med. Biol. 527 , 705-12, (2003) To study the complex inter-relationships between inflammatory and apoptotic responses and the kynurenine pathway, we have utilized electron-capture negative ion mass spectrometry to develop trace anal... |
| Dihydrobaikiaine |
| UNII:H254GW7PVV |
| (±)-Piperidine-2-carboxylic acid |
| DL-Pipecolinic acid |
| DL-Pipecolinicacid |
| Pipecolinic acid |
| 6-CARBOXYPIPERIDINE |
| DL-2-Piperidinecarboxylic acid |
| (±)-Pipecolinic acid |
| Pipecolic acid |
| α-Pipecolinic acid |
| .α.-Pipecolinic acid |
| a-Pipecolinic acid |
| (±)-Pipecolic acid |
| 2-Carboxypiperidine |
| piperidine-2-carboxylic acid |
| DL-Homoproline |
| DL-Pipecolic Acid |
| MFCD00064347 |
| (±)-2-Piperidinecarboxylic acid |
| EINECS 208-616-3 |
| 2-Piperidinecarboxylic acid |
| DL-PIPECOLINIC ACID:PIPERIDINE-2-CARBOXYLIC ACID |
 CAS#:18650-39-0
CAS#:18650-39-0
