metyrapone
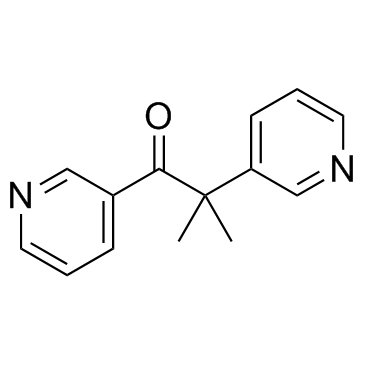
metyrapone structure
|
Common Name | metyrapone | ||
|---|---|---|---|---|
| CAS Number | 54-36-4 | Molecular Weight | 226.274 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 384.4±22.0 °C at 760 mmHg | |
| Molecular Formula | C14H14N2O | Melting Point | 53-56ºC | |
| MSDS | Chinese USA | Flash Point | 189.3±28.8 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of metyraponeMetyrapone is an inhibitor of cytochrome P450-mediated ω/ω-1 hydroxylase activity and CYP11B1.Target: CYP11B1Metyrapone is a drug used in the diagnosis of adrenal insufficiency and occasionally in the treatment of Cushing's syndrome (hypercortisolism). Metyrapone blocks cortisol synthesis by reversibly inhibiting steroid 11β-hydroxylase. This stimulates ACTH secretion, which in turn increases plasma 11-Deoxycortisol levels. |
| Name | metyrapone |
|---|---|
| Synonym | More Synonyms |
| Description | Metyrapone is an inhibitor of cytochrome P450-mediated ω/ω-1 hydroxylase activity and CYP11B1.Target: CYP11B1Metyrapone is a drug used in the diagnosis of adrenal insufficiency and occasionally in the treatment of Cushing's syndrome (hypercortisolism). Metyrapone blocks cortisol synthesis by reversibly inhibiting steroid 11β-hydroxylase. This stimulates ACTH secretion, which in turn increases plasma 11-Deoxycortisol levels. |
|---|---|
| Related Catalog |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 384.4±22.0 °C at 760 mmHg |
| Melting Point | 53-56ºC |
| Molecular Formula | C14H14N2O |
| Molecular Weight | 226.274 |
| Flash Point | 189.3±28.8 °C |
| Exact Mass | 226.110611 |
| PSA | 42.85000 |
| LogP | 1.19 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.565 |
| Storage condition | Store at +4°C |
| Water Solubility | H2O: soluble (sparingly) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319-H335 |
| Precautionary Statements | P301 + P312 + P330-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn |
| Risk Phrases | 22-36/37/38 |
| Safety Phrases | 26 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UC3050000 |
| HS Code | 2933399090 |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Cytochrome P450-dependent metabolism of caffeine in Drosophila melanogaster.
PLoS ONE 10(2) , e0117328, (2015) Caffeine (1, 3, 7-trimethylxanthine), an alkaloid produced by plants, has antioxidant and insecticide properties that can affect metabolism and cognition. In vertebrates, the metabolites derived from ... |
|
|
The different roles of glucocorticoids in the hippocampus and hypothalamus in chronic stress-induced HPA axis hyperactivity.
PLoS ONE 9(5) , e97689, (2014) Hypothalamus-pituitary-adrenal (HPA) hyperactivity is observed in many patients suffering from depression and the mechanism underling the dysfunction of HPA axis is not well understood. Chronic stress... |
|
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental... |
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Inhibition of cytochrome P450 in Sprague-Dawley rat liver microsomes assessed as inhi...
Source: ChEMBL
Target: N/A
External Id: CHEMBL4420723
|
|
Name: Evaluated for inhibition of bovine adrenal cortical mitochondrial 11 beta-hydroxylase
Source: ChEMBL
Target: Cytochrome P450 11B1, mitochondrial
External Id: CHEMBL615228
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
|
Name: uHTS identification of cystic fibrosis induced NFkb Inhibitors in a fluoresence assay
Source: Burnham Center for Chemical Genomics
Target: cystic fibrosis transmembrane conductance regulator [Homo sapiens]
External Id: SBCCG-A764-CF-PAF-Primary-Assay
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ant...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_ANT_FLUO8_1536_1X%INH PRUN
|
| Metopyrone |
| Metopiron |
| Chlorisondamine diiodide |
| Mepyrapone |
| Metyrapone |
| Methopirapone |
| Metroprione |
| Methapyrapone |
| Metyrapon |
| Metopirone |
| Methopyrinine |
| Methopyrone |
| 2-methyl-1,2-dipyridin-3-ylpropan-1-one |
| 2-Methyl-1,2-di(pyridin-3-yl)propan-1-one |
| Methopyrapone |
| 2-Methyl-1,2-di(3-pyridinyl)-1-propanone |
| 2-méthyl-1,2-dipyridin-3-ylpropan-1-one |
| 2-Methyl-1,2-dipyridin-3-ylpropan-1-on |
 CAS#:15825-89-5
CAS#:15825-89-5 CAS#:6304-18-3
CAS#:6304-18-3 CAS#:15031-77-3
CAS#:15031-77-3 CAS#:17159-42-1
CAS#:17159-42-1
