H-Ser-Ome·HCl
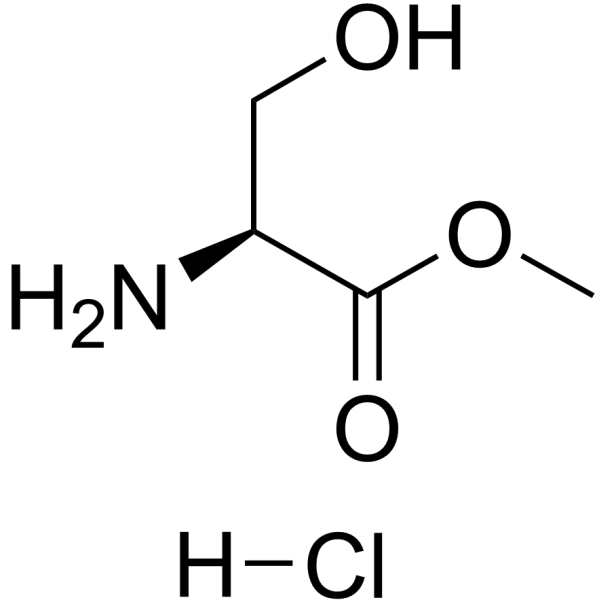
H-Ser-Ome·HCl structure
|
Common Name | H-Ser-Ome·HCl | ||
|---|---|---|---|---|
| CAS Number | 5680-80-8 | Molecular Weight | 155.580 | |
| Density | N/A | Boiling Point | 234.7ºC at 760 mmHg | |
| Molecular Formula | C4H10ClNO3 | Melting Point | 160-165ºC | |
| MSDS | Chinese USA | Flash Point | 95.8ºC | |
Use of H-Ser-Ome·HClL-Serine methyl ester (hydrochloride) is a serine derivative[1]. |
| Name | L-Serine methyl ester hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | L-Serine methyl ester (hydrochloride) is a serine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Boiling Point | 234.7ºC at 760 mmHg |
|---|---|
| Melting Point | 160-165ºC |
| Molecular Formula | C4H10ClNO3 |
| Molecular Weight | 155.580 |
| Flash Point | 95.8ºC |
| Exact Mass | 155.034927 |
| PSA | 72.55000 |
| InChIKey | NDBQJIBNNUJNHA-DFWYDOINSA-N |
| SMILES | COC(=O)C(N)CO.Cl |
| Storage condition | -20°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 29225000 |
|
~98% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: Tetrahedron, , vol. 50, # 31 p. 9411 - 9424 |
|
~87% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: Journal of Organic Chemistry, , vol. 55, # 8 p. 2271 - 2272 |
|
~99% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: Chemical Communications, , vol. 46, # 3 p. 445 - 447 |
|
~% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: Tetrahedron Letters, , vol. 54, # 48 p. 6511 - 6513 |
|
~% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: US5113009 A1, ; |
|
~% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: US5523463 A1, ; |
|
~% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: Tetrahedron Letters, , vol. 29, # 2 p. 235 - 238 |
|
~% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: Synthesis, , # 19 art. no. P06210SS, p. 3353 - 3357 |
|
~% 
H-Ser-Ome·HCl CAS#:5680-80-8 |
| Literature: Synthesis, , # 19 art. no. P06210SS, p. 3353 - 3357 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2922509090 |
|---|---|
| Summary | 2922509090. other amino-alcohol-phenols, amino-acid-phenols and other amino-compounds with oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Transport and signaling via the amino acid binding site of the yeast Gap1 amino acid transceptor.
Nat. Chem. Biol. 5 , 45-52, (2009) Transporter-related nutrient sensors, called transceptors, mediate nutrient activation of signaling pathways through the plasma membrane. The mechanism of action of transporting and nontransporting tr... |
|
|
Acylated serine derivatives: a unique class of arthropod pheromones of the Australian redback spider, Latrodectus hasselti.
Angew. Chem. Int. Ed. Engl. 49(11) , 2037-40, (2010)
|
|
|
The novel and efficient direct synthesis of N,O-acetal compounds using a hypervalent iodine(III) reagent: an improved synthetic method for a key intermediate of discorhabdins.
Chem. Commun. (Camb.) (13) , 1764-6, (2005) The use of hypervalent iodine(III) reagents allowed us to develop the novel and efficient direct synthesis of N,O-acetal compounds via the oxidative fragmentation reaction of alpha-amino acids or alph... |
|
Name: Inhibition of nitrogen-starved wild type sigma1278b yeast Gap1-mediated amino acid up...
Source: ChEMBL
Target: General amino-acid permease GAP1
External Id: CHEMBL1228071
|
| L-Serine, methyl ester, hydrochloride (1:1) |
| MFCD00063680 |
| methyl (2S)-2-amino-3-hydroxypropanoate,hydrochloride |
| DL-Serine, methyl ester, hydrochloride |
| Methyl L-serinate hydrochloride (1:1) |
| methyl DL-serinate hydrochloride |
| EINECS 227-140-7 |
| Q1YZVO1 &&HCl |
| L-Serine, methyl ester, hydrochloride |
| Serine, methyl ester, hydrochloride (1:1) |
| Methyl serinate hydrochloride |
| Methyl serinate hydrochloride (1:1) |
| DL-serine methylester Hydrochloride |
| H-Ser-OMe·HCl |

![(2S)-methyl 2-(2-azabicyclo[2.2.1]hept-5-en-2-yl)-3-hydroxypropanoate structure](https://image.chemsrc.com/caspic/330/111934-17-9.png)

![tert-butyl 1-({[(1S)-1-(hydroxymethyl)-2-methoxy-2-oxoethyl]amino}carbonyl)-(1S,4R)-7-azabicyclo[2.2.1]hept-2-ene-7-carboxylic acid structure](https://image.chemsrc.com/caspic/301/1258511-48-6.png)

![tert-butyl 1-({[(1S)-1-(hydroxymethyl)-2-methoxy-2-oxoethyl]amino}carbonyl)-(1R,4S)-7-azabicyclo[2.2.1]hept-2-ene-7-carboxylic acid structure](https://image.chemsrc.com/caspic/339/1258511-47-5.png)
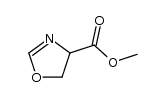
 CAS#:1065102-64-8
CAS#:1065102-64-8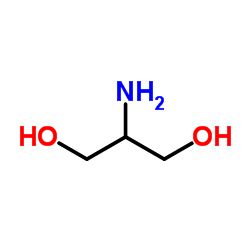 CAS#:534-03-2
CAS#:534-03-2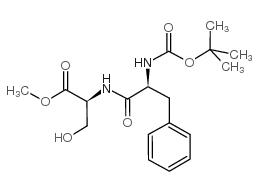 CAS#:34290-59-0
CAS#:34290-59-0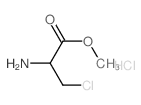 CAS#:17136-54-8
CAS#:17136-54-8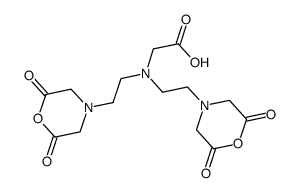 CAS#:2311-26-4
CAS#:2311-26-4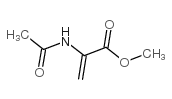 CAS#:35356-70-8
CAS#:35356-70-8 CAS#:81629-69-8
CAS#:81629-69-8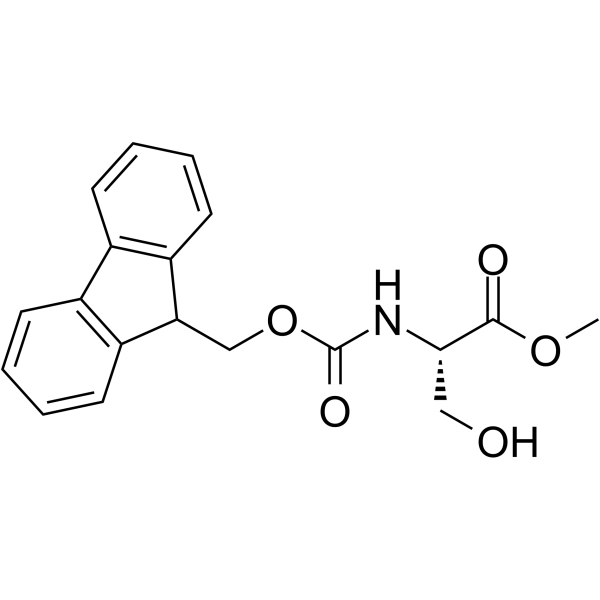 CAS#:82911-78-2
CAS#:82911-78-2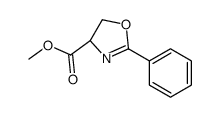 CAS#:78715-83-0
CAS#:78715-83-0 CAS#:88099-67-6
CAS#:88099-67-6
