alpha-Tocopherol acetate
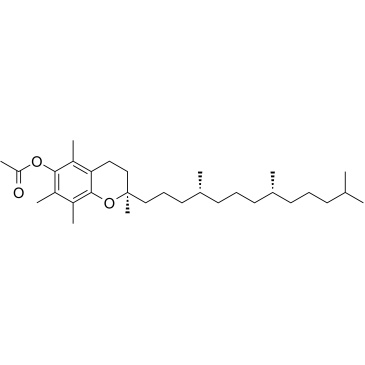
alpha-Tocopherol acetate structure
|
Common Name | alpha-Tocopherol acetate | ||
|---|---|---|---|---|
| CAS Number | 58-95-7 | Molecular Weight | 472.743 | |
| Density | 0.9±0.1 g/cm3 | Boiling Point | 184 ºC | |
| Molecular Formula | C31H52O3 | Melting Point | 28 ºC | |
| MSDS | Chinese USA | Flash Point | 235.6±24.7 °C | |
Use of alpha-Tocopherol acetateD-α-Tocopherol acetate (D-Vitamin E acetate) can be hydrolyzed to d-alpha-tocopherol (VE) and absorbed in the small intestine[1]. |
| Name | D-alpha-Tocopheryl acetate |
|---|---|
| Synonym | More Synonyms |
| Description | D-α-Tocopherol acetate (D-Vitamin E acetate) can be hydrolyzed to d-alpha-tocopherol (VE) and absorbed in the small intestine[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 0.9±0.1 g/cm3 |
|---|---|
| Boiling Point | 184 ºC |
| Melting Point | 28 ºC |
| Molecular Formula | C31H52O3 |
| Molecular Weight | 472.743 |
| Flash Point | 235.6±24.7 °C |
| Exact Mass | 472.391632 |
| PSA | 35.53000 |
| LogP | 12.07 |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.488 |
| InChIKey | ZAKOWWREFLAJOT-CEFNRUSXSA-N |
| SMILES | CC(=O)Oc1c(C)c(C)c2c(c1C)CCC(C)(CCCC(C)CCCC(C)CCCC(C)C)O2 |
| Water Solubility | <0.1 g/100 mL at 17 ºC |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves |
|---|---|
| Safety Phrases | S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 1 |
| RTECS | GP8280000 |
| Precursor 7 | |
|---|---|
| DownStream 5 | |
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental... |
|
|
Phytantriol-based in situ liquid crystals with long-term release for intra-articular administration.
AAPS PharmSciTech 16 , 846-54, (2015) The purpose of this study was to develop an injectable in situ liquid crystal formulation for intra-articular (IA) administration, and in situ forming a viscous liquid crystalline gel with long-term r... |
|
|
Microbeads: a novel multiparticulate drug delivery technology for increasing the solubility and dissolution of celecoxib.
Pharm. Dev. Technol. 20(2) , 211-8, (2015) The purpose of this study was to develop a novel multipaticulate drug delivery technology suitable for the delivery of pre-solubilized celecoxib to the gastrointestinal tract and more specifically to ... |
| (2R)-2,5,7,8-Tetramethyl-2-[(4R,8R)-4,8,12-trimethyltridecyl]-3,4-dihydro-2H-chromen-6-yl acetate |
| a-Tocopheryl Acetate |
| Vitamin E acetate |
| [2R*(4R*,8R*)]-3,4-Dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-2H-1-benzopyran-6-ol Acetate |
| EINECS 231-710-0 |
| Eprolin |
| Tocopheryl acetate |
| E-Vimin |
| Evion |
| MFCD00072052 |
| Vitamin- E acetate |
| D-α-Tocopheryl acetate |
| a-Tocopherol Acetate |
| TOCOPHEROL ACETATE |
 CAS#:505-32-8
CAS#:505-32-8 CAS#:11113-50-1
CAS#:11113-50-1 CAS#:108-24-7
CAS#:108-24-7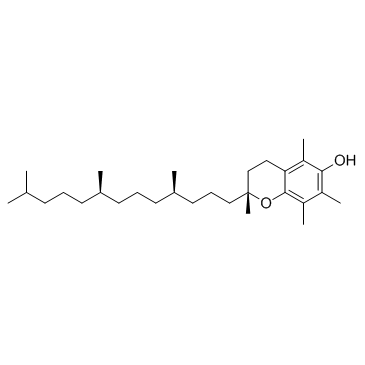 CAS#:59-02-9
CAS#:59-02-9 CAS#:2140-82-1
CAS#:2140-82-1 CAS#:64-19-7
CAS#:64-19-7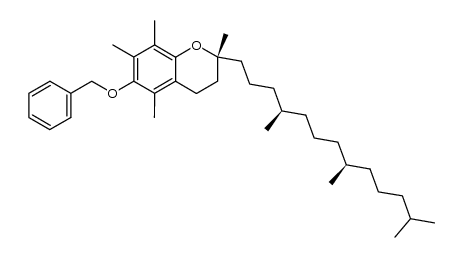 CAS#:59965-06-9
CAS#:59965-06-9 CAS#:7559-04-8
CAS#:7559-04-8 CAS#:14745-36-9
CAS#:14745-36-9
