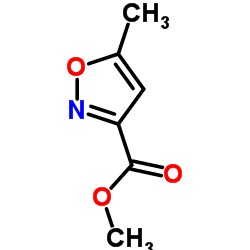
Isocarboxazid

Isocarboxazid structure
|
Common Name | Isocarboxazid | ||
|---|---|---|---|---|
| CAS Number | 59-63-2 | Molecular Weight | 231.251 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 394.5±42.0 °C at 760 mmHg | |
| Molecular Formula | C12H13N3O2 | Melting Point | 98-100ºC | |
| MSDS | N/A | Flash Point | 192.4±27.9 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of IsocarboxazidIsocarboxazid is a non-selective and irreversible inhibitor of monoamine oxidase, with an IC50 of 4.8 μM for rat brain monoamine oxidase in vitro[1]. |
| Name | N'-benzyl-5-methyl-1,2-oxazole-3-carbohydrazide |
|---|---|
| Synonym | More Synonyms |
| Description | Isocarboxazid is a non-selective and irreversible inhibitor of monoamine oxidase, with an IC50 of 4.8 μM for rat brain monoamine oxidase in vitro[1]. |
|---|---|
| Related Catalog | |
| Target |
IC50: 4.8 μM (rat brain monoamine oxidase)[1]. |
| In Vivo | Isocarboxazid (1, 3 mg/kg, i.p., 60 min) pretreatment in mice shows the significant increased number of head twitches at 15 and 30 min after 5-HTP[2]. Isocarboxazid (1, 3 mg/kg, i.p., 60 min) treatment in mice together with 5-HTP administration causes 43% 5-HT concentration increased and 22% of 5-HIAA decreased compared to brain concentrations in mice given 5-HTP alone[2]. Animal Model: Twelve male mice of dd strain (20-25 g)[2] Dosage: 0, 0.3,1, 3 mg/kg Administration: Intraperitoneally 60 min before intravenous injection of 5-HTP Result: The number of head twitches at 15 and 30 min after 5-HTP was increased. 43% 5-HT concentration increased and 22% of 5-HIAA decreased. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 394.5±42.0 °C at 760 mmHg |
| Melting Point | 98-100ºC |
| Molecular Formula | C12H13N3O2 |
| Molecular Weight | 231.251 |
| Flash Point | 192.4±27.9 °C |
| Exact Mass | 231.100784 |
| PSA | 67.16000 |
| LogP | 1.03 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.573 |
| InChIKey | XKFPYPQQHFEXRZ-UHFFFAOYSA-N |
| SMILES | Cc1cc(C(=O)NNCc2ccccc2)no1 |
| Storage condition | -20°C Freezer |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | Missing Phrase - N15.00950417 |
| RIDADR | UN 3249 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2934999090 |
|
~% 
Isocarboxazid CAS#:59-63-2 |
| Literature: Journal of Heterocyclic Chemistry, , vol. 46, # 5 p. 909 - 913 |
|
~% 
Isocarboxazid CAS#:59-63-2 |
| Literature: Journal of Medicinal and Pharmaceutical Chemistry, , vol. 2, p. 133 - 145 |
|
~% 
Isocarboxazid CAS#:59-63-2 |
| Literature: Journal of Medicinal and Pharmaceutical Chemistry, , vol. 2, p. 133 - 145 |
|
~% 
Isocarboxazid CAS#:59-63-2 |
| Literature: Journal of Medicinal and Pharmaceutical Chemistry, , vol. 2, p. 133 - 145 |
|
~% 
Isocarboxazid CAS#:59-63-2 |
| Literature: Journal of Medicinal and Pharmaceutical Chemistry, , vol. 2, p. 133 - 145 US2908688 , ; |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Dicer-mediated maturation of pre-microRNA
Source: Center for Chemical Genomics, University of Michigan
Target: N/A
External Id: TargetID_659_CEMA
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization-based biochemical high throughput primary assay to identify...
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id: SIAE_INH_FP_1536_1X%INH PRUN
|
|
Name: MITF Measured in Cell-Based System Using Plate Reader - 2084-01_Activator_SinglePoint...
Source: Broad Institute
Target: N/A
External Id: 2084-01_Activator_SinglePoint_HTS_Activity
|
|
Name: Counterscreen for inhibitors of the fructose-bisphosphate aldolase (FBA) of M. tuberc...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: GDH-TPI_INH_ABS_1536_1X%INH CSRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of Cac...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303805
|
|
Name: Inhibition of cell viability relative to arbidol control (inhibition index > 1 indica...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303819
|
| isocarboxazide |
| Benazide |
| 5-Methyl-isoxazol-3-carbonsaeure-(N'-benzyl-hydrazid) |
| Marplan |
| Isocarboxazidum |
| N'-Benzyl-5-methylisoxazol-3-carbohydrazid |
| Isocarboxazida |
| 3-Isoxazolecarboxylic acid, 5-methyl-, 2-(phenylmethyl)hydrazide |
| N'-benzyl-5-méthylisoxazole-3-carbohydrazide |
| Isocarbonazid |
| Isocarboxazid |
| N'-benzyl-5-methylisoxazole-3-carbohydrazide |
| N-Benzyl-N'-<5-methyl-isoxazol-3-yl>-carbonyl-hydrazin |
| Isocarbossazide |
| N'-Benzyl-5-methyl-1,2-oxazole-3-carbohydrazide |
| Isocarboxyzid |
| Enerzer |
| 5-methyl-isoxazole-3-carboxylic acid-(N'-benzyl-hydrazide) |
| isocarboxacid |
| 5-methyl-N'-(phenylmethyl)isoxazole-3-carbohydrazide |


 CAS#:491594-42-4
CAS#:491594-42-4