Megestrol acetate
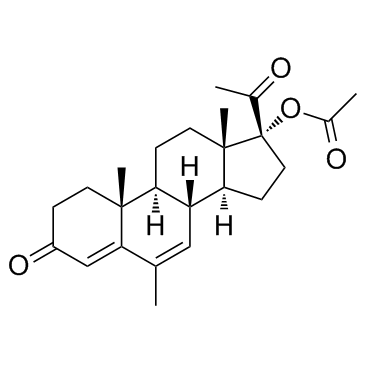
Megestrol acetate structure
|
Common Name | Megestrol acetate | ||
|---|---|---|---|---|
| CAS Number | 595-33-5 | Molecular Weight | 384.509 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 507.1±50.0 °C at 760 mmHg | |
| Molecular Formula | C24H32O4 | Melting Point | 214°C | |
| MSDS | Chinese USA | Flash Point | 218.5±30.2 °C | |
| Symbol |

GHS08 |
Signal Word | Warning | |
Use of Megestrol acetateMegestrol Acetate is a synthetic progesteronal agent with an IC50 of 260 μM for the inhibition of HegG2.Target: Progesterone ReceptorMegestrol acetate, also known as 17α-acetoxy-6-dehydro-6-methylprogesterone, and sometimes abbreviated as MGA or MA, is a steroidal progestin and progesterone derivative (specifically, a 17-hydroxylated progesterone) with predominantly progestational and antigonadotropic effects. Megestrol acetate is a good candidate for muscle wasting treatment and future studies addressed at the interaction between the drug and protein turnover in human skeletal muscle should be performed. |
| Name | megestrol acetate |
|---|---|
| Synonym | More Synonyms |
| Description | Megestrol Acetate is a synthetic progesteronal agent with an IC50 of 260 μM for the inhibition of HegG2.Target: Progesterone ReceptorMegestrol acetate, also known as 17α-acetoxy-6-dehydro-6-methylprogesterone, and sometimes abbreviated as MGA or MA, is a steroidal progestin and progesterone derivative (specifically, a 17-hydroxylated progesterone) with predominantly progestational and antigonadotropic effects. Megestrol acetate is a good candidate for muscle wasting treatment and future studies addressed at the interaction between the drug and protein turnover in human skeletal muscle should be performed. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 507.1±50.0 °C at 760 mmHg |
| Melting Point | 214°C |
| Molecular Formula | C24H32O4 |
| Molecular Weight | 384.509 |
| Flash Point | 218.5±30.2 °C |
| Exact Mass | 384.230072 |
| PSA | 60.44000 |
| LogP | 3.82 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.551 |
| InChIKey | RQZAXGRLVPAYTJ-GQFGMJRRSA-N |
| SMILES | CC(=O)OC1(C(C)=O)CCC2C3C=C(C)C4=CC(=O)CCC4(C)C3CCC21C |
| Storage condition | Refrigerator |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H351-H373 |
| Precautionary Statements | P281 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful |
| Risk Phrases | R40;R48 |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | TU4075000 |
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental... |
|
|
Translating clinical findings into knowledge in drug safety evaluation--drug induced liver injury prediction system (DILIps).
J. Sci. Ind. Res. 65(10) , 808, (2006) Drug-induced liver injury (DILI) is a significant concern in drug development due to the poor concordance between preclinical and clinical findings of liver toxicity. We hypothesized that the DILI typ... |
|
|
Multi-target spectral moment QSAR versus ANN for antiparasitic drugs against different parasite species.
Bioorg. Med. Chem. 18 , 2225-31, (2010) There are many of pathogen parasite species with different susceptibility profile to antiparasitic drugs. Unfortunately, almost QSAR models predict the biological activity of drugs against only one pa... |
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Increase the activity of the Burkholderia fixLJ 2-component system
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: Burkholderia multivorans
External Id: HMS1625
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
|
Name: uHTS identification of cystic fibrosis induced NFkb Inhibitors in a fluoresence assay
Source: Burnham Center for Chemical Genomics
Target: cystic fibrosis transmembrane conductance regulator [Homo sapiens]
External Id: SBCCG-A764-CF-PAF-Primary-Assay
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ant...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_ANT_FLUO8_1536_1X%INH PRUN
|
| (8R,9S,10R,13S,14S,17R)-17-Acetyl-6,10,13-trimethyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-ylacetat |
| Megestil |
| Pregna-4,6-diene-3,20-dione, 17-hydroxy-6-methyl-, acetate |
| Megestin |
| Pruritex Chat |
| Felipil |
| Nuvacon |
| 6-Dehydro-6-methyl-17a-acetoxyprogesterone |
| Citestrol |
| Megace |
| Megoestrolacetat |
| Megostat |
| 6-Methyl-3,20-dioxopregna-4,6-dien-17-yl acetate |
| Megestrol acetate |
| Megestrol acetate [USAN] |
| 17α-Acetoxy-6-methylpregna-4,6-diene-3,20-dione |
| Megestrol 17-acetate |
| Pill'Kan |
| Estropill |
| Kombiquens |
| Megefren |
| EINECS 209-864-5 |
| 17α-Acetoxy-6-methyl-4,6-pregnadiene-3,20-dione |
| Maygace |
| Volidan |
| 6-Methyl-D4,6-pregnadien-17a-ol-3,20-dione Acetate |
| 17a-Acetoxy-6-methylpregna-4,6-diene-3,20-dione |
| Minigest |
| Megestryl acetate |
| Nia |
| acétate de (8R,9S,10R,13S,14S,17R)-17-acétyl-6,10,13-triméthyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodécahydro-1H-cyclopenta[a]phénanthrén-17-yle |
| 6-Dehydro-6-methyl-17α-acetoxyprogesterone |
| Opochaleurs |
| Suppress |
| [(8R,9S,10R,13S,14S,17R)-17-acetyl-6,10,13-trimethyl-3-oxo-2,8,9,11,12,14,15,16-octahydro-1H-cyclopenta[a]phenanthren-17-yl] acetate |
| Megecat |
| Volplan |
| Progesterone, 6-dehydro-17-hydroxy-6-methyl-, acetate |
| Pilucalm |
| MFCD00056470 |
| Gorda |
| Canipil |
| (8R,9S,10R,13S,14S,17R)-17-acetyl-6,10,13-trimethyl-3-oxo-2,3,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate |
| Megestrol (Acetate) |
 CAS#:32634-95-0
CAS#:32634-95-0 CAS#:4382-76-7
CAS#:4382-76-7![(8R,9S,10R,13S,14S,17R)-17-acetyl-10,13-dimethyl-3-oxo-2,3,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl acetate Structure](https://image.chemsrc.com/caspic/386/4134-58-1.png) CAS#:4134-58-1
CAS#:4134-58-1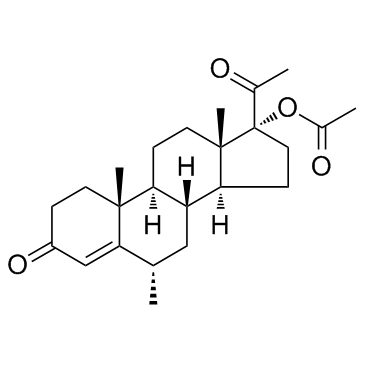 CAS#:71-58-9
CAS#:71-58-9 CAS#:1863-39-4
CAS#:1863-39-4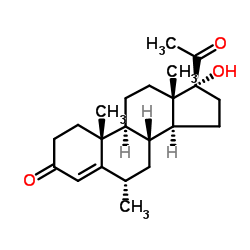 CAS#:520-85-4
CAS#:520-85-4 CAS#:113665-92-2
CAS#:113665-92-2 CAS#:3562-63-8
CAS#:3562-63-8
