Fluorescein Diacetate
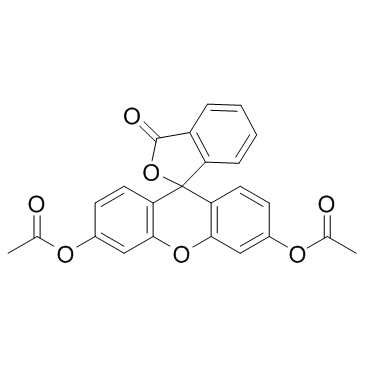
Fluorescein Diacetate structure
|
Common Name | Fluorescein Diacetate | ||
|---|---|---|---|---|
| CAS Number | 596-09-8 | Molecular Weight | 416.380 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 604.7±55.0 °C at 760 mmHg | |
| Molecular Formula | C24H16O7 | Melting Point | 200-203 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 264.0±31.5 °C | |
Use of Fluorescein DiacetateFluorescein diacetate is a cell permeable esterase-substrate. Fluorescein diacetate can be used as a fluorogenic substrate for hGSTP1-1. |
| Name | Fluorescein Diacetate |
|---|---|
| Synonym | More Synonyms |
| Description | Fluorescein diacetate is a cell permeable esterase-substrate. Fluorescein diacetate can be used as a fluorogenic substrate for hGSTP1-1. |
|---|---|
| Related Catalog | |
| In Vitro | Fluorescein diacetate (FDA) is an acetylated derivative of the green fluorescent dye fluorescein[1]. Fluorescein diacetate (FDA), a fluorescent probe used for vital staining, is a fluorescently activated by esterolytic activity of human Pi-class glutathione S-transferase (hGSTP1) selectively among various cytosolic GSTs. Fluorescence activation of Fluorescein diacetate susceptible to GST inhibitors is observed in MCF7 cells exogenously overexpressing hGSTP1, but not in cells overexpressing hGSTA1 or hGSTM1. Fluorescein diacetate can be used as a fluorogenic substrate for hGSTP1-1. To investigate whether the fluorescence activation is due to hGSTP1 activity, Fluorescein diacetate is incubated with recombinant hGSTP1-1 and GSH in vitro. Remarkable fluorescence activation is observed in the presence of both hGSTP1-1 and GSH, whereas only slight activation is observed in the absence of either of them or when the enzyme is heat inactivated. This suggests that the fluorescence activation of Fluorescein diacetate depends on hGSTP1-1 activity. From the linear relationship between the rate of increase in fluorescence and the hGSTP1-1 concentration, the specific activity of the enzyme for 1 μM Fluorescein diacetate is determined to be 79±15 nmol/min/mg protein. Fluorescein diacetate is applicable as a fluorogenic substrate for evaluating inhibitors of GSTP1-1 in vitro. For Fluorescein diacetate as a substrate, both Ethacrynic acid (EA) and NBDHEX suppress the hGSTP1-1-dependent fluorescent increase in a concentration-dependent manner, with IC50s of 3.3±0.5 μM and 0.61±0.04 μM, respectively[2]. |
| Cell Assay | MCF7 cells (2-3×105) are seeded in a 35-mm glass bottom dish before the experiment. Prior to imaging, cells are washed with 1 mL PBS, then incubated in 1 mL Hanks’ Balanced Salt solution (HBSS(+) without phenol red) containing 1 μM Fluorescein diacetate (0.1% DMSO as a cosolvent) for 5 min at 37°C. Cells are washed twice with 1 mL PBS and 1 mL HBSS is added before imaging. Fluorescence images of fluorescein and DsRed-Express2 were acquired in the FITC channel (excitation at 473 nm) and the DsRed channel (excitation 559 nm). The 16-bit images obtained are analysed[2]. |
| References |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 604.7±55.0 °C at 760 mmHg |
| Melting Point | 200-203 °C(lit.) |
| Molecular Formula | C24H16O7 |
| Molecular Weight | 416.380 |
| Flash Point | 264.0±31.5 °C |
| Exact Mass | 416.089600 |
| PSA | 88.13000 |
| LogP | 2.77 |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.681 |
| InChIKey | CHADEQDQBURGHL-UHFFFAOYSA-N |
| SMILES | CC(=O)Oc1ccc2c(c1)Oc1cc(OC(C)=O)ccc1C21OC(=O)c2ccccc21 |
| Storage condition | −20°C |
|
~72% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: WO2008/73764 A2, ; Page/Page column 23 ; |
|
~% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Journal of the Association of Official Agricultural Chemists, , vol. 34, p. 114,115 |
|
~% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Tetrahedron Letters, , vol. 44, # 52 p. 9359 - 9362 |
|
~% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Tetrahedron Letters, , vol. 44, # 52 p. 9359 - 9362 |
|
~% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Journal of the American Chemical Society, , vol. 30, p. 862 |
|
~% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Justus Liebigs Annalen der Chemie, , vol. 183, p. 31 Justus Liebigs Annalen der Chemie, , vol. 372, p. 109 |
|
~93% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Dyes and Pigments, , vol. 89, # 2 p. 120 - 126 |
|
~% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Chemische Berichte, , vol. 7, p. 1212 Justus Liebigs Annalen der Chemie, , vol. 183, p. 31 Justus Liebigs Annalen der Chemie, , vol. 372, p. 109 |
|
~% 
Fluorescein Dia... CAS#:596-09-8 |
| Literature: Chemische Berichte, , vol. 7, p. 1212 Justus Liebigs Annalen der Chemie, , vol. 183, p. 31 Justus Liebigs Annalen der Chemie, , vol. 372, p. 109 |
| Precursor 9 | |
|---|---|
| DownStream 1 | |
|
Preconditioning L6 Muscle Cells with Naringin Ameliorates Oxidative Stress and Increases Glucose Uptake.
PLoS ONE 10 , e0132429, (2015) Enhanced oxidative stress contributes to pathological changes in diabetes and its complications. Thus, strategies to reduce oxidative stress may alleviate these pathogenic processes. Herein, we have i... |
|
|
Chondrogenesis of human bone marrow mesenchymal stromal cells in highly porous alginate-foams supplemented with chondroitin sulfate.
Mater. Sci. Eng. C. Mater. Biol. Appl. 50 , 160-72, (2015) To overcome the limited intrinsic cartilage repair, autologous chondrocyte or bone-marrow-derived mesenchymal stromal cell (BM-MSC) was implanted into cartilage defects. For this purpose suitable bioc... |
|
|
Dewetting based fabrication of fibrous micro-scaffolds as potential injectable cell carriers.
Mater. Sci. Eng. C. Mater. Biol. Appl. 48 , 663-72, (2015) Although regenerative medicine utilizing tissue scaffolds has made enormous strides in recent years, many constraints still hamper their effectiveness. A limitation of many scaffolds is that they form... |
|
Name: Inhibition of SARS-CoV-2-induced cytopathy was measured 4 days after infection of EGF...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL5303712
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Absorbance-based primary biochemical high throughput screening assay to identify acti...
Source: The Scripps Research Institute Molecular Screening Center
Target: caspase-3 preproprotein [Homo sapiens]
External Id: PROCASPASE3_ACT_EPIABS_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule modulators of NR3A
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: SBCCG-A1015-NR3A-Primary-Assay
|
|
Name: MITF Measured in Cell-Based System Using Plate Reader - 2084-01_Activator_SinglePoint...
Source: Broad Institute
Target: N/A
External Id: 2084-01_Activator_SinglePoint_HTS_Activity
|
| EINECS 209-877-6 |
| 3',6'-Bis(acetyloxy)spiro(isobenzofuran-1(3H),9'-(9H)xanthen)-3-one |
| 3-Oxospiro(isobenzofuran-1(3H),9'-(9H)xanthene)-3',6'-diyl diacetate |
| MFCD00005062 |
| 3-Oxo-3H-spiro[2-benzofuran-1,9'-xanthene]-3',6'-diyl diacetate |
| (6'-acetyloxy-3-oxospiro[2-benzofuran-1,9'-xanthene]-3'-yl) acetate |
| Fluorescein Diacetate |


![3-oxo-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-3H-spiro[isobenzofuran-1,9'-xanthene]-3',6'-diyl diacetate structure](https://image.chemsrc.com/caspic/417/649734-78-1.png)