Psoralen
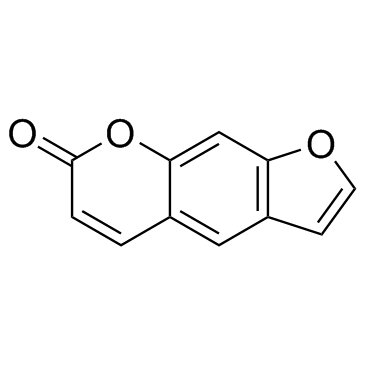
Psoralen structure
|
Common Name | Psoralen | ||
|---|---|---|---|---|
| CAS Number | 66-97-7 | Molecular Weight | 186.163 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 362.6±27.0 °C at 760 mmHg | |
| Molecular Formula | C11H6O3 | Melting Point | 160-162 °C | |
| MSDS | Chinese USA | Flash Point | 173.1±23.7 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of PsoralenPsoralen(Furocoumarin) is an active ingredient from Fructus Psoraleae; has anticancer activity.IC50 value:Target:in vitro: Psoralen dosages of 1-10 μM exhibited low cytotoxicity toward chondrocytes. However, a dosage of 100 μM suppressed the proliferation of chondrocytes. Different concentrations of psoralen treatments on chondrocytes revealed that GAG and Type II collagen synthesis increased, especially at 100 μM, by 0.39-fold and 0.48-fold, respectively, on day 3, and by 0.51-fold and 0.56-fold, respectively, on day 9 [1]. in vivo: Tumor volume inhibition rates were 43.75% and 40.18%, respectively, in the psoralen and isopsoralen low-dose groups, and tumor weight inhibition rates were 38.83% and 37.77%. Tumor volume inhibition rates were 67.86% and 66.96%, respectively, in the psoralen and isopsoralen high-dose groups, and tumor weight inhibition rates were 49.47% and 47.87% [2]. psoralen can inhibit metastasis of breast cancer to bone in vivo. Histological, molecular biological, and imaging analyses revealed that psoralen inhibits bone metastases in mice [3]. |
| Name | psoralen |
|---|---|
| Synonym | More Synonyms |
| Description | Psoralen(Furocoumarin) is an active ingredient from Fructus Psoraleae; has anticancer activity.IC50 value:Target:in vitro: Psoralen dosages of 1-10 μM exhibited low cytotoxicity toward chondrocytes. However, a dosage of 100 μM suppressed the proliferation of chondrocytes. Different concentrations of psoralen treatments on chondrocytes revealed that GAG and Type II collagen synthesis increased, especially at 100 μM, by 0.39-fold and 0.48-fold, respectively, on day 3, and by 0.51-fold and 0.56-fold, respectively, on day 9 [1]. in vivo: Tumor volume inhibition rates were 43.75% and 40.18%, respectively, in the psoralen and isopsoralen low-dose groups, and tumor weight inhibition rates were 38.83% and 37.77%. Tumor volume inhibition rates were 67.86% and 66.96%, respectively, in the psoralen and isopsoralen high-dose groups, and tumor weight inhibition rates were 49.47% and 47.87% [2]. psoralen can inhibit metastasis of breast cancer to bone in vivo. Histological, molecular biological, and imaging analyses revealed that psoralen inhibits bone metastases in mice [3]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 362.6±27.0 °C at 760 mmHg |
| Melting Point | 160-162 °C |
| Molecular Formula | C11H6O3 |
| Molecular Weight | 186.163 |
| Flash Point | 173.1±23.7 °C |
| Exact Mass | 186.031693 |
| PSA | 43.35000 |
| LogP | 1.67 |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.667 |
| InChIKey | ZCCUUQDIBDJBTK-UHFFFAOYSA-N |
| SMILES | O=c1ccc2cc3ccoc3cc2o1 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATAMUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | LV0944000 |
| Precursor 9 | |
|---|---|
| DownStream 4 | |
|
Rotavirus increases levels of lipidated LC3 supporting accumulation of infectious progeny virus without inducing autophagosome formation.
PLoS ONE 9(4) , e95197, (2014) Replication of many RNA viruses benefits from subversion of the autophagic pathway through many different mechanisms. Rotavirus, the main etiologic agent of pediatric gastroenteritis worldwide, has be... |
|
|
Inhibition of human aldehyde oxidase activity by diet-derived constituents: structural influence, enzyme-ligand interactions, and clinical relevance.
Drug Metab. Dispos. 43(1) , 34-41, (2014) The mechanistic understanding of interactions between diet-derived substances and conventional medications in humans is nascent. Most investigations have examined cytochrome P450-mediated interactions... |
|
|
Assessment of a candidate marker constituent predictive of a dietary substance-drug interaction: case study with grapefruit juice and CYP3A4 drug substrates.
J. Pharmacol. Exp. Ther. 351(3) , 576-84, (2014) Dietary substances, including herbal products and citrus juices, can perpetrate interactions with conventional medications. Regulatory guidances for dietary substance-drug interaction assessment are l... |
|
Name: Inhibition of SARS-CoV-2-induced cytopathy was measured 4 days after infection of EGF...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL5303712
|
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Inhibition of mouse brain monoamine oxidase
Source: ChEMBL
Target: N/A
External Id: CHEMBL2380137
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Inhibition of NF-KB p50 subunit/DNA interaction after 20 mins by EMSA
Source: ChEMBL
Target: Nuclear factor NF-kappa-B p105 subunit
External Id: CHEMBL1291967
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Inhibition of human recombinant MAO-B expressed in baculovirus infected BTI insect ce...
Source: ChEMBL
Target: Amine oxidase [flavin-containing] B
External Id: CHEMBL4424159
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
| Furocoumarin |
| 7H-Furo[3,2-g]chromen-7-one |
| Psoralen |
| EINECS 200-639-7 |
| Psoralene |
| prosuler |
| 6,7-Furanocoumarin |
| MFCD00010520 |
| Psorline-P |
| FICUSIN |
| 7H-Furo[3,2-g][1]benzopyran-7-one |
 CAS#:849144-95-2
CAS#:849144-95-2 CAS#:6544-89-4
CAS#:6544-89-4 CAS#:7535-48-0
CAS#:7535-48-0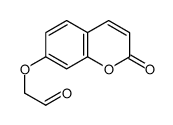 CAS#:16851-02-8
CAS#:16851-02-8![7-oxo-2,3-dihydro-7H-furo[3,2-g]chromene-6-carboxylic acid Structure](https://image.chemsrc.com/caspic/378/52656-95-8.png) CAS#:52656-95-8
CAS#:52656-95-8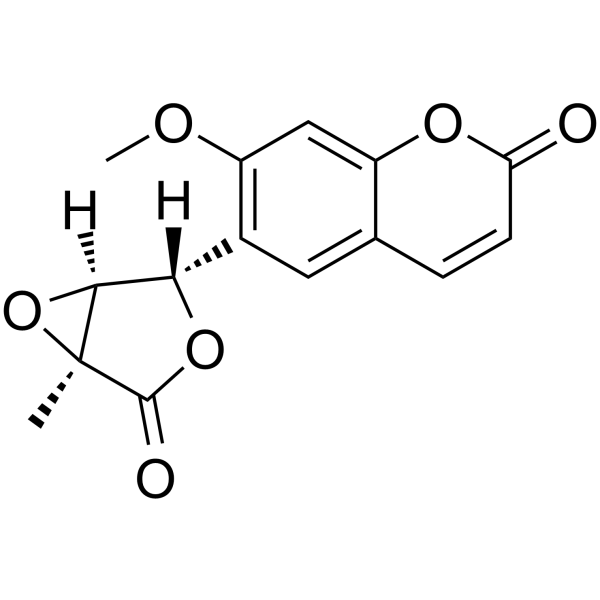 CAS#:15085-71-9
CAS#:15085-71-9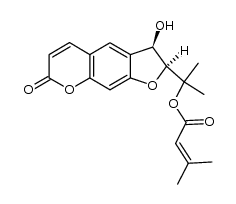 CAS#:287171-02-2
CAS#:287171-02-2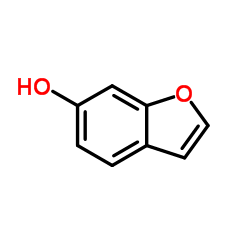 CAS#:13196-11-7
CAS#:13196-11-7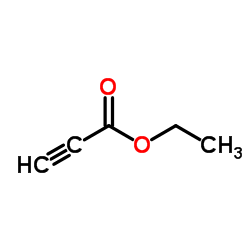 CAS#:623-47-2
CAS#:623-47-2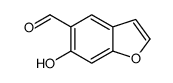 CAS#:20073-22-7
CAS#:20073-22-7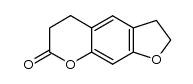 CAS#:19881-52-8
CAS#:19881-52-8
