Drospirenone
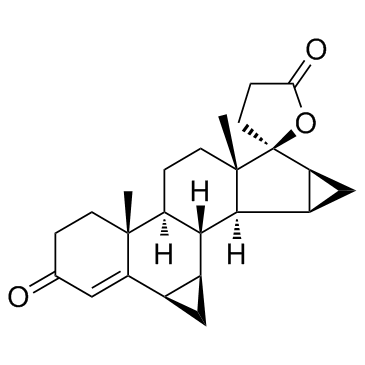
Drospirenone structure
|
Common Name | Drospirenone | ||
|---|---|---|---|---|
| CAS Number | 67392-87-4 | Molecular Weight | 366.493 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 552.2±50.0 °C at 760 mmHg | |
| Molecular Formula | C24H30O3 | Melting Point | 196-200ºC | |
| MSDS | Chinese USA | Flash Point | 241.6±30.2 °C | |
| Symbol |

GHS08 |
Signal Word | Danger | |
Use of DrospirenoneDrospirenone(Dihydrospirorenone) is a synthetic progestin that is an analog to spironolactone.Target: Progesterone ReceptorDrospirenone is a novel progestin under clinical development that is similar to the natural hormone progesterone, combining potent progestogenic with antimineralocorticoid and antiandrogenic activities. drospirenone was devoid of glucocorticoid activity. Both progestins did not show any antiglucocorticoid action. Furthermore, drospirenone and progesterone both showed considerable antimineralocorticoid activity and weak mineralocorticoid activity [1]. the pharmacological profile of drospirenone is more closely related to that of the natural hormone progesterone than is that of any other synthetic progestogen in use today. Therefore, drospirenone is anticipated to give rise to a number of additional health benefits both for users of oral contraceptives and hormone replacement therapy recipients [2]. The combination of 17beta-estradiol and drospirenone has a positive effect on BMD and a potentially beneficial effect on lipids. Although endometrial thickness increased slightly, the safety of the endometrium was assured, as no cases of hyperplasia or cancer occurred [3].Clinical indications: Acne; Dysmenorrhea; Endometriosis; Female contraception; Folic acid deficiency; Premenstrual syndrome |
| Name | drospirenone |
|---|---|
| Synonym | More Synonyms |
| Description | Drospirenone(Dihydrospirorenone) is a synthetic progestin that is an analog to spironolactone.Target: Progesterone ReceptorDrospirenone is a novel progestin under clinical development that is similar to the natural hormone progesterone, combining potent progestogenic with antimineralocorticoid and antiandrogenic activities. drospirenone was devoid of glucocorticoid activity. Both progestins did not show any antiglucocorticoid action. Furthermore, drospirenone and progesterone both showed considerable antimineralocorticoid activity and weak mineralocorticoid activity [1]. the pharmacological profile of drospirenone is more closely related to that of the natural hormone progesterone than is that of any other synthetic progestogen in use today. Therefore, drospirenone is anticipated to give rise to a number of additional health benefits both for users of oral contraceptives and hormone replacement therapy recipients [2]. The combination of 17beta-estradiol and drospirenone has a positive effect on BMD and a potentially beneficial effect on lipids. Although endometrial thickness increased slightly, the safety of the endometrium was assured, as no cases of hyperplasia or cancer occurred [3].Clinical indications: Acne; Dysmenorrhea; Endometriosis; Female contraception; Folic acid deficiency; Premenstrual syndrome |
|---|---|
| Related Catalog | |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 552.2±50.0 °C at 760 mmHg |
| Melting Point | 196-200ºC |
| Molecular Formula | C24H30O3 |
| Molecular Weight | 366.493 |
| Flash Point | 241.6±30.2 °C |
| Exact Mass | 366.219482 |
| PSA | 43.37000 |
| LogP | 3.15 |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Index of Refraction | 1.610 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H360 |
| Precautionary Statements | P201-P280-P308 + P313 |
| Safety Phrases | 36/37 |
| RIDADR | NONH for all modes of transport |
| RTECS | WH1299000 |
| HS Code | 2937290090 |
| HS Code | 2937290090 |
|---|
|
Postural tachycardia syndrome (POTS).
Circulation 127(23) , 2336-42, (2013)
|
|
|
Sexual function, mood and menopause symptoms in Lithuanian postmenopausal women.
Climacteric 16(1) , 185-93, (2013) To assess sexual function in a clinical sample of Lithuanian postmenopausal women and identify the most important determinants of sexual function, including the use of hormone replacement therapy (HT)... |
|
|
Liquid chromatography with diode array detection and multivariate curve resolution for the selective and sensitive quantification of estrogens in natural waters.
Anal. Chim. Acta 835 , 19-28, (2014) Following the green analytical chemistry principles, an efficient strategy involving second-order data provided by liquid chromatography (LC) with diode array detection (DAD) was applied for the simul... |
| (1aR,5aR,5bS,7aS,8S,8aS,9aS,9bS,9cR,9dR)-5a,7a-Dimethyl-1,1a,5,5a,5b,6,7,7a,8a,9,9a,9b,9c,9d-tetradecahydro-3'H-spiro[cyclopropa[4,5]cyclopenta[1,2-a]cyclopropa[l]phenanthren-8,2'-furan]-3,5'(4H,4'H)- |
| (1aR,5aR,5bS,7aS,8S,8aS,9aS,9bS,9cR,9dR)-5a,7a-Dimethyl-1,1a,5,5a,5b,6,7,7a,8a,9,9a,9b,9c,9d-tetradecahydro-3'H-spiro[cyclopropa[4,5]cyclopenta[1,2-a]cyclopropa[l]phenanthren-8,2'-furan]-3,5'(4H,4'H)-dion |
| arbolactone |
| spiro[8H-cyclopropa[4,5]cyclopenta[1,2-a]cyclopropa[l]phenanthrene-8,2'(5'H)-furan]-3,5'(4H)-dione, 1,1a,3',4',5,5a,5b,6,7,7a,8a,9,9a,9b,9c,9d-hexadecahydro-5a,7a-dimethyl-, (1aR,5aR,5bS,7aS,8S,8aS,9a |
| 6b,7b:15b,16b-Dimethylene-3-oxo-4-androstene-[17(b-1')-spiro-5']perhydrofuran-2'-one |
| Spiro[8H-cyclopropa[4,5]cyclopenta[1,2-a]cyclopropa[l]phenanthrene-8,2'(5'H)-furan]-3,5'(4H)-dione, 1,1a,3',4',5,5a,5b,6,7,7a,8a,9,9a,9b,9c,9d-hexadecahydro-5a,7a-dimethyl-, (1aR,5aR,5bS,7aS,8S,8aS,9aS,9bS,9cR,9dR)- |
| Drospirenona |
| Drospirenone |
| Drospirenon |
| 6b,7b |
| Dorospirenone |
| (1aR,5aR,5bS,7aS,8S,8aS,9aS,9bS,9cR,9dR)-5a,7a-Dimethyl-1,1a,5,5a,5b,6,7,7a,8a,9,9a,9b,9c,9d-tetradecahydro-3'H-spiro[cyclopropa[4,5]cyclopenta[1,2-a]cyclopropa[l]phenanthrene-8,2'-furan]-3,5'(4H,4'H)-dione |
| 1,2-Dihydrospirorenone |
| ZK 3059 |
| (1aR,5aR,5bS,7aS,8S,8aS,9aS,9bS,9cR,9dR)-5a,7a-diméthyl-1,1a,5,5a,5b,6,7,7a,8a,9,9a,9b,9c,9d-tétradécahydro-3'H-spiro[cyclopropa[4,5]cyclopenta[1,2-a]cyclopropa[l]phénanthrène-8,2'-furane]-3,5'(4H,4'H)-dione |
| (1aR,5aR,5bS,7aS,8S,8aS,9aS,9bS,9cR,9dR)-5a,7a-dimethyl-1,1a,5,5a,5b,6,7,7a,8a,9,9a,9b,9c,9d-tetradecahydro-3'H-spiro[cyclopropa[4,5]cyclopenta[1,2-a]cyclopropa[l]phenanthrene-8,2'-furan]-3,5'(4H,4'H) |
| YASMIN |
| dihydrospirorenone |

