Lidocaine hydrochloride
Modify Date: 2025-08-20 17:57:33

Lidocaine hydrochloride structure
|
Common Name | Lidocaine hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 73-78-9 | Molecular Weight | 270.798 | |
| Density | N/A | Boiling Point | 350.8ºC at 760 mmHg | |
| Molecular Formula | C14H23ClN2O | Melting Point | 80-82°C | |
| MSDS | N/A | Flash Point | 166ºC | |
Use of Lidocaine hydrochlorideLidocaine Hcl salt, an amide local anesthetic, has anti-inflammatory properties in vitro and in vivo, possibly due to an attenuation of pro-inflammatory cytokines, intracellular adhesion molecule-1 (ICAM-1), and reduction of neutrophils influx.Target: Lidocaine is a common local anesthetic and antiarrhythmic drug. Lidocaine is used topically to relieve itching, burning and pain from skin inflammations, injected as a dental anesthetic or as a local anesthetic for minor surgery. Lidocaine, the first amino amide–type local anesthetic, was first synthesized under the name xylocaine by Swedish chemist Nils Lofgren in 1943. His colleague Bengt Lundqvist performed the first injection anesthesia experiments on himself.Lidocaine is approximately 95% metabolized (dealkylated) in the liver by CYP3A4 to the pharmacologically-active metabolites monoethylglycinexylidide (MEGX) and then subsequently to the inactive glycine xylidide. MEGX has a longer half life than lidocaine but also is a less potent sodium channel blocker. The elimination half-life of lidocaine is approximately 90–120 minutes in most patients. This may be prolonged in patients with hepatic impairment (average 343 minutes) or congestive heart failure (average 136 minutes). |
| Name | lidocaine hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Description | Lidocaine Hcl salt, an amide local anesthetic, has anti-inflammatory properties in vitro and in vivo, possibly due to an attenuation of pro-inflammatory cytokines, intracellular adhesion molecule-1 (ICAM-1), and reduction of neutrophils influx.Target: Lidocaine is a common local anesthetic and antiarrhythmic drug. Lidocaine is used topically to relieve itching, burning and pain from skin inflammations, injected as a dental anesthetic or as a local anesthetic for minor surgery. Lidocaine, the first amino amide–type local anesthetic, was first synthesized under the name xylocaine by Swedish chemist Nils Lofgren in 1943. His colleague Bengt Lundqvist performed the first injection anesthesia experiments on himself.Lidocaine is approximately 95% metabolized (dealkylated) in the liver by CYP3A4 to the pharmacologically-active metabolites monoethylglycinexylidide (MEGX) and then subsequently to the inactive glycine xylidide. MEGX has a longer half life than lidocaine but also is a less potent sodium channel blocker. The elimination half-life of lidocaine is approximately 90–120 minutes in most patients. This may be prolonged in patients with hepatic impairment (average 343 minutes) or congestive heart failure (average 136 minutes). |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 350.8ºC at 760 mmHg |
|---|---|
| Melting Point | 80-82°C |
| Molecular Formula | C14H23ClN2O |
| Molecular Weight | 270.798 |
| Flash Point | 166ºC |
| Exact Mass | 270.149902 |
| PSA | 32.34000 |
| LogP | 3.45870 |
| InChIKey | IYBQHJMYDGVZRY-UHFFFAOYSA-N |
| SMILES | CCN(CC)CC(=O)Nc1c(C)cccc1C.Cl |
| Storage condition | Refrigerator |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| RIDADR | 3249 |
|---|---|
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2924299090 |
| Precursor 0 | |
|---|---|
| DownStream 2 | |
| HS Code | 2924299090 |
|---|---|
| Summary | 2924299090. other cyclic amides (including cyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Inhibition of neurosphere proliferation of mouse neural precursor cells by MTT assay
Source: ChEMBL
Target: N/A
External Id: CHEMBL1266185
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Flow Cytometric HTS Screen for inhibitors of the ABC transporter ABCB6 for Validation...
Source: NMMLSC
Target: ATP-binding cassette sub-family B member 6, mitochondrial [Homo sapiens]
External Id: UNMCMD_ABCB6_1o_ValidationSet
|
|
Name: Identifying Sarm1 Tir Hydrolase inhibitors through NAD-Glo assay
Source: 24386
Target: N/A
External Id: Sarm1 Tir NADase inhibitors screen
|
Total 184, Current Page 1 of 19
1
2
3
4
5
| LIDOCAINE HCL |
| 2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide,hydrochloride |
| 2-(Diethylamino)-N-(2,6-dimethylphenyl)acetamidhydrochlorid |
| Xyloneural |
| XYLOCAINE HYDROCHLORIDE |
| N-(2,6-Dimethylphenyl)-N,N-diethylglycinamide hydrochloride (1:1) |
| MFCD00034865 |
| (Diethylamino)-2',6'-acetoxylidide Monohydrochloride |
| 2-(diéthylamino)-N-(2,6-diméthylphényl)acétamide chlorhydrate |
| Xilina hydrochloride |
| 2-(Diethylamino)-N-(2,6-dimethylphenyl)acetamide, hydrochloride (1:1) |
| N-(2,6-dimethylphenyl)-N,N-diethylglycinamide hydrochloride |
| Lidocaine hydrochloride |
| lignocaine hydrochloride |
| 2-(Diethylamino)-N-(2,6-dimethylphenyl)acetamide monohydrochloride |
| Acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, hydrochloride (1:1) |
| Lidocaton |
| Lidothesin |
| Laryng-O-jet |
| N-(2,6-dimethylphenyl)-N2,N2-diethylglycinamide hydrochloride |
| 2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide hydrochloride |
| EINECS 200-803-8 |
| 2',6'-Acetoxylidide, 2-(diethylamino)-, monohydrochloride (8CI) |
| Xylocard |
| 2N2&1VMR B1 F1 &&HCl |
| Lidocaine (hydrochloride) |
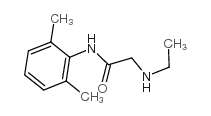 CAS#:7728-40-7
CAS#:7728-40-7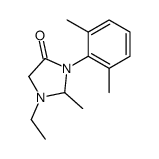 CAS#:32845-42-4
CAS#:32845-42-4