Schisandrol A
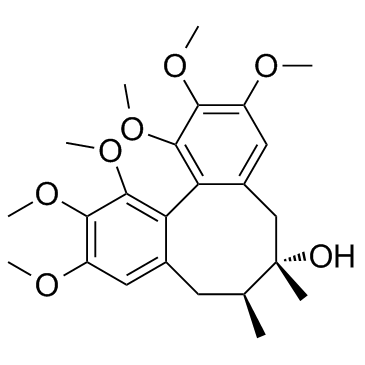
Schisandrol A structure
|
Common Name | Schisandrol A | ||
|---|---|---|---|---|
| CAS Number | 7432-28-2 | Molecular Weight | 432.507 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 576.7±50.0 °C at 760 mmHg | |
| Molecular Formula | C24H32O7 | Melting Point | 128-129ºC | |
| MSDS | Chinese USA | Flash Point | 302.6±30.1 °C | |
| Symbol |


GHS07, GHS09 |
Signal Word | Warning | |
Use of Schisandrol ASchisandrin has various therapeutic effects on a range of medical conditions such as anti-asthmatic, anti-cancer, and anti-inflammatory effects.IC50 value:Target:in vitro: Sch inhibited the pro-fibrotic activity of TGF-β1 in AML12 cells; thus, it suppressed the accumulation of ECM proteins. Also, Sch inhibited the EMT as assessed by reduced expression of vimentin and fibronectin, and increased E-cadherin and ZO-1 in TGF-β1 induced AML12 cells. Sch reduced TGF-β1-mediated phosphorylation of Smad2/3 and Smad3/4 DNA binding activity. On the other hand, Sch reduced TGF-β1-induced ERK1/2 and PI3K/Akt phosphorylation in the non-Smad pathway [1]. the anti-inflammatory properties of schisandrin result from the inhibition of nitric oxide (NO) production, prostaglandin E(2) (PGE(2)) release, cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) expression, which in turn results from the inhibition of nuclear factor-kappaB (NF-kappaB), c-Jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinase (MAPK) activities in a RAW 264.7 macrophage cell line [2]. |
| Name | Schisandrin |
|---|---|
| Synonym | More Synonyms |
| Description | Schisandrin has various therapeutic effects on a range of medical conditions such as anti-asthmatic, anti-cancer, and anti-inflammatory effects.IC50 value:Target:in vitro: Sch inhibited the pro-fibrotic activity of TGF-β1 in AML12 cells; thus, it suppressed the accumulation of ECM proteins. Also, Sch inhibited the EMT as assessed by reduced expression of vimentin and fibronectin, and increased E-cadherin and ZO-1 in TGF-β1 induced AML12 cells. Sch reduced TGF-β1-mediated phosphorylation of Smad2/3 and Smad3/4 DNA binding activity. On the other hand, Sch reduced TGF-β1-induced ERK1/2 and PI3K/Akt phosphorylation in the non-Smad pathway [1]. the anti-inflammatory properties of schisandrin result from the inhibition of nitric oxide (NO) production, prostaglandin E(2) (PGE(2)) release, cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) expression, which in turn results from the inhibition of nuclear factor-kappaB (NF-kappaB), c-Jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinase (MAPK) activities in a RAW 264.7 macrophage cell line [2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 576.7±50.0 °C at 760 mmHg |
| Melting Point | 128-129ºC |
| Molecular Formula | C24H32O7 |
| Molecular Weight | 432.507 |
| Flash Point | 302.6±30.1 °C |
| Exact Mass | 432.214813 |
| PSA | 75.61000 |
| LogP | 4.18 |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.535 |
| InChIKey | YEFOAORQXAOVJQ-PUCHIXIPSA-N |
| SMILES | COc1cc2c(c(OC)c1OC)-c1c(cc(OC)c(OC)c1OC)CC(C)(O)C(C)C2 |
| Storage condition | room temp |
| Water Solubility | DMSO: ≥13mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |


GHS07, GHS09 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H400 |
| Precautionary Statements | P301 + P312 + P330 |
| Hazard Codes | Xn,N |
| Risk Phrases | 22-50/53 |
| Safety Phrases | 60-61 |
| RIDADR | UN 3077 9 / PGIII |
| Hazard Class | 9.0 |
| HS Code | 2932999099 |
| Precursor 0 | |
|---|---|
| DownStream 2 | |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Effects of acupuncture at ST36 on pharmacokinetics of Schisandra lignans in rats.
Acupunct. Med. 33 , 223-9, (2015) To investigate the influence of acupuncture at ST36 on the pharmacokinetics of Schisandra lignans including schisandrin, deoxyschisandrin and schisandrin B after intragastric administration of Schisan... |
|
|
Comparative pharmacokinetics and tissue distribution profiles of lignan components in normal and hepatic fibrosis rats after oral administration of Fuzheng Huayu recipe.
J. Ethnopharmacol. 166 , 305-12, (2015) Fuzheng Huayu recipe (FZHY) is formulated on the basis of Chinese medicine theory in treating liver fibrosis.To illuminate the influence of the pathological state of liver fibrosis on the pharmacokine... |
|
|
The Lignan-containing Extract of Schisandra chinensis Berries Inhibits the Growth of Chlamydia pneumonia.
Nat. Prod. Commun. 10 , 1001-4, (2015) The purpose of this study was to investigate the effect and selectivity of an extract of Schisandra chinensis berries against Chlamydia pneumoniae and C. trachomatis. Among the ethnopharmacological us... |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Antioxidant activity in human HL60 cells assessed as inhibition of TPA-stimulated hyd...
Source: ChEMBL
Target: HL-60
External Id: CHEMBL977837
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Cytotoxicity against human BT549 cells upto 48 uM
Source: ChEMBL
Target: BT-549
External Id: CHEMBL977843
|
|
Name: Cytotoxicity against human KB cells upto 48 uM
Source: ChEMBL
Target: KB
External Id: CHEMBL977842
|
|
Name: Antiinflammatory activity against LPS-induced prostaglandin E2 production in mouse ma...
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL977845
|
|
Name: Cytotoxicity against human SKOV3 cells upto 48 uM
Source: ChEMBL
Target: SK-OV-3
External Id: CHEMBL977844
|
| Schizandrin |
| Schisandrol A |
| schizandra |
| 1,2,3,10,11,12-Hexamethoxy-6,7-dimethyl-5,6,7,8-tetrahydrodibenzo[a,c][8]annulen-6-ol |
| Schisandrol |
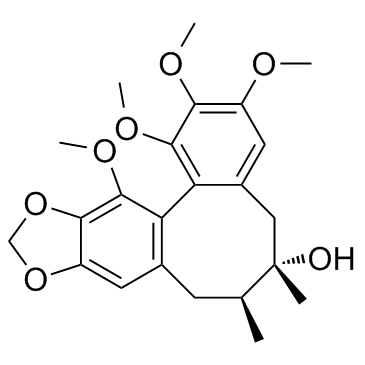 CAS#:58546-54-6
CAS#:58546-54-6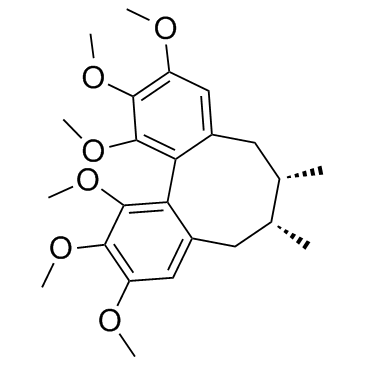 CAS#:61281-38-7
CAS#:61281-38-7
