isopropylamine
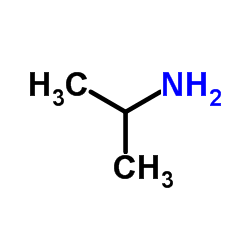
isopropylamine structure
|
Common Name | isopropylamine | ||
|---|---|---|---|---|
| CAS Number | 75-31-0 | Molecular Weight | 59.110 | |
| Density | 0.7±0.1 g/cm3 | Boiling Point | 30.9±8.0 °C at 760 mmHg | |
| Molecular Formula | C3H9N | Melting Point | -101 °C | |
| MSDS | Chinese USA | Flash Point | -32.2±0.0 °C | |
| Symbol |



GHS02, GHS05, GHS06 |
Signal Word | Danger | |
| Name | isopropylamine |
|---|---|
| Synonym | More Synonyms |
| Density | 0.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 30.9±8.0 °C at 760 mmHg |
| Melting Point | -101 °C |
| Molecular Formula | C3H9N |
| Molecular Weight | 59.110 |
| Flash Point | -32.2±0.0 °C |
| Exact Mass | 59.073498 |
| PSA | 26.02000 |
| LogP | 0.21 |
| Vapour density | 2.04 (vs air) |
| Vapour Pressure | 607.7±0.1 mmHg at 25°C |
| Index of Refraction | 1.389 |
| InChIKey | JJWLVOIRVHMVIS-UHFFFAOYSA-N |
| SMILES | CC(C)N |
| Stability | Stable. Extremely flammable - note low boiling point and low flash point. Readily forms explosive mixtures with air. Incompatible with strong oxidizing agents, acids, acid chlorides, acid anhydrides, perchloryl fluoride. |
| Water Solubility | soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |



GHS02, GHS05, GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H224-H301 + H311 + H331-H314-H335 |
| Precautionary Statements | P210-P261-P280-P303 + P361 + P353-P305 + P351 + P338-P370 + P378 |
| Personal Protective Equipment | Faceshields;full-face respirator (US);Gloves;Goggles;multi-purpose combination respirator cartridge (US) |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R12;R36/37/38 |
| Safety Phrases | S16-S26-S29 |
| RIDADR | UN 1221 3/PG 1 |
| WGK Germany | 1 |
| RTECS | NT8400000 |
| Packaging Group | I |
| Hazard Class | 3 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
|
Comparison of microwave, ultrasound and accelerated-assisted solvent extraction for recovery of polyphenols from Citrus sinensis peels.
Food Chem. 187 , 507-16, (2015) Peel of Citrus sinensis contains significant amounts of bioactive polyphenols that could be used as ingredients for a number of value-added products with health benefits. Extraction of polyphenols fro... |
|
|
Evolution of strategies to achieve baseline separation of ten anionic, water-soluble sulfated estrogens via achiral packed column supercritical fluid chromatography.
J. Chromatogr. A. 1370 , 240-5, (2014) Near baseline separation of ten sulfated sodium salts of various structurally related estrogens employing a variety of bonded stationary phase packed columns was obtained using a conventional supercri... |
|
|
Generic chiral method development in supercritical fluid chromatography and ultra-performance supercritical fluid chromatography.
J. Chromatogr. A. 1363 , 311-22, (2014) The development of chiral separation methods in pharmaceutical industry is often a very tedious, labour intensive and expensive process. A trial-and-error approach remains frequently used, given the u... |
| Dimethylmethaneamine |
| 1-Methylethylamine |
| 2-Propanamine |
| Isopropylamine |
| 2-Aminopropane |
| MFCD00008082 |
| 2-Amino-propaan |
| propan-2-amine |
| isopropyl amine |
| 2-Propylamine |
| 2-AMINO-PROPANE |
| sec-Propylamine |
| EINECS 200-860-9 |
| Propane,2-amino |
| MONOISOPROPYLAMINE |
 CAS#:79-46-9
CAS#:79-46-9 CAS#:187737-37-7
CAS#:187737-37-7 CAS#:56-81-5
CAS#:56-81-5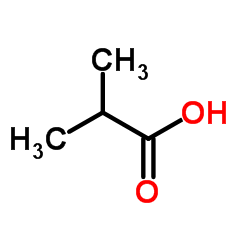 CAS#:79-31-2
CAS#:79-31-2 CAS#:67-64-1
CAS#:67-64-1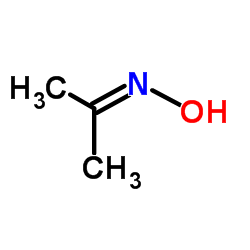 CAS#:127-06-0
CAS#:127-06-0 CAS#:103-02-6
CAS#:103-02-6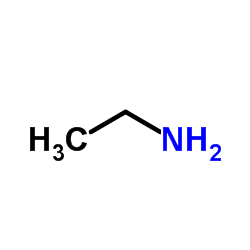 CAS#:75-04-7
CAS#:75-04-7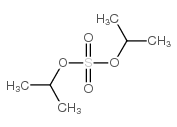 CAS#:2973-10-6
CAS#:2973-10-6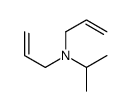 CAS#:10542-47-9
CAS#:10542-47-9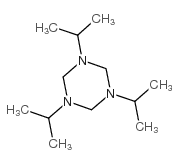 CAS#:10556-98-6
CAS#:10556-98-6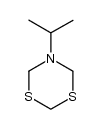 CAS#:34866-41-6
CAS#:34866-41-6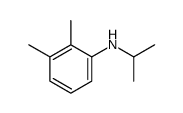 CAS#:63114-76-1
CAS#:63114-76-1 CAS#:105336-21-8
CAS#:105336-21-8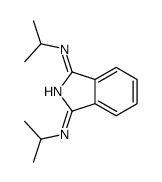 CAS#:104830-26-4
CAS#:104830-26-4 CAS#:106500-93-0
CAS#:106500-93-0 CAS#:104678-18-4
CAS#:104678-18-4![2-methyl-N-[phenyl-(propan-2-ylamino)phosphoryl]propan-2-amine structure](https://image.chemsrc.com/caspic/366/111783-67-6.png) CAS#:111783-67-6
CAS#:111783-67-6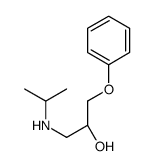 CAS#:106351-44-4
CAS#:106351-44-4
