D-(-)-Quinic acid
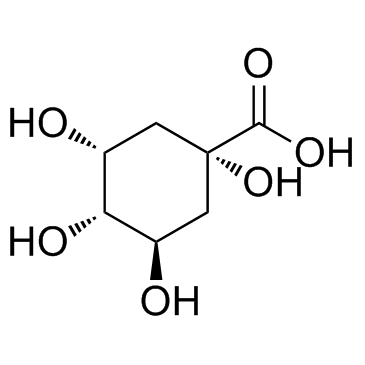
D-(-)-Quinic acid structure
|
Common Name | D-(-)-Quinic acid | ||
|---|---|---|---|---|
| CAS Number | 77-95-2 | Molecular Weight | 192.167 | |
| Density | 1.8±0.1 g/cm3 | Boiling Point | 438.4±45.0 °C at 760 mmHg | |
| Molecular Formula | C7H12O6 | Melting Point | 165-170 °C | |
| MSDS | Chinese USA | Flash Point | 233.1±25.2 °C | |
Use of D-(-)-Quinic acidD-(-)-Quinic acid is a cyclohexanecarboxylic acid and is implicated in the perceived acidity of coffee. |
| Name | Quinic acid |
|---|---|
| Synonym | More Synonyms |
| Description | D-(-)-Quinic acid is a cyclohexanecarboxylic acid and is implicated in the perceived acidity of coffee. |
|---|---|
| Related Catalog |
| Density | 1.8±0.1 g/cm3 |
|---|---|
| Boiling Point | 438.4±45.0 °C at 760 mmHg |
| Melting Point | 165-170 °C |
| Molecular Formula | C7H12O6 |
| Molecular Weight | 192.167 |
| Flash Point | 233.1±25.2 °C |
| Exact Mass | 192.063385 |
| PSA | 118.22000 |
| LogP | -2.01 |
| Vapour Pressure | 0.0±2.4 mmHg at 25°C |
| Index of Refraction | 1.687 |
| InChIKey | AAWZDTNXLSGCEK-LNVDRNJUSA-N |
| SMILES | O=C(O)C1(O)CC(O)C(O)C(O)C1 |
| Water Solubility | 400 g/l (20 ºC) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | GU8650000 |
| HS Code | 29181980 |
| Precursor 5 | |
|---|---|
| DownStream 10 | |
| HS Code | 29181980 |
|---|
|
Antioxidant properties, phenolic composition and potentiometric sensor array evaluation of commercial and new blueberry (Vaccinium corymbosum) and bog blueberry (Vaccinium uliginosum) genotypes.
Food Chem. 188 , 583-90, (2015) Antioxidant properties of juices of newly bred and known blueberry (Vaccinium corymbosum) genotypes and wild bog blueberry (Vaccinium uliginosum) were evaluated by ABTS(+) scavenging capacity (RSC), f... |
|
|
Investigating the potential of under-utilised plants from the Asteraceae family as a source of natural antimicrobial and antioxidant extracts.
Food Chem. 161 , 79-86, (2014) Antimicrobial properties of ethanol and water extracts from eight Asteraceae species were investigated against three Gram positive (Staphylococcus aureus, MRSA and Bacillus cereus) and two Gram negati... |
|
|
Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression.
Nature 457(7231) , 910-4, (2009) Multiple, complex molecular events characterize cancer development and progression. Deciphering the molecular networks that distinguish organ-confined disease from metastatic disease may lead to the i... |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Fluorescence polarization-based biochemical high throughput primary assay to identify...
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=Sialate O-acetylesterase; AltName: Full=H-Lse; AltName: Full=Sialic acid-specific 9-O-acetylesterase; Flags: Precursor [Homo sapiens]
External Id: SIAE_INH_FP_1536_1X%INH PRUN
|
|
Name: uHTS identification of small molecule modulators of NR3A
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: SBCCG-A1015-NR3A-Primary-Assay
|
|
Name: uHTS identification of small molecule modulators of Rev-erb Alpha.
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: SBCCG-A1016-RevErbaLBD-Primary-Assay
|
| (1R,3R,4R,5R)-(-)-quinic acid |
| Kinicacid |
| (1S,3R,4s)-1,3,4,5-Tetrahydroxycyclohexanecarboxylic acid |
| [1R-(1a,3a,4a,5b)]-1,3,4,5-Tetrahydroxycyclohexanecarboxylic Acid |
| CHINIC ACID |
| l-Chinic acid |
| D-(-)-quinic acid |
| 5beta)]-alph |
| EINECS 201-072-8 |
| (-)-quinic acid |
| Chinasaure |
| MFCD00003864 |
| l-Kinic acid |
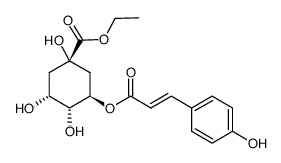 CAS#:1443678-33-8
CAS#:1443678-33-8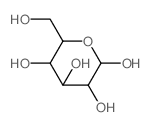 CAS#:2280-44-6
CAS#:2280-44-6 CAS#:67-56-1
CAS#:67-56-1 CAS#:138-59-0
CAS#:138-59-0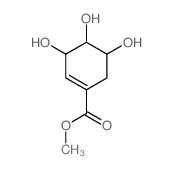 CAS#:40983-58-2
CAS#:40983-58-2 CAS#:124-38-9
CAS#:124-38-9 CAS#:111936-95-9
CAS#:111936-95-9 CAS#:7732-18-5
CAS#:7732-18-5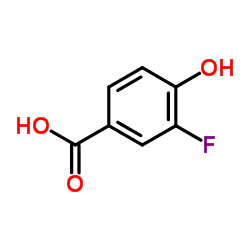 CAS#:350-29-8
CAS#:350-29-8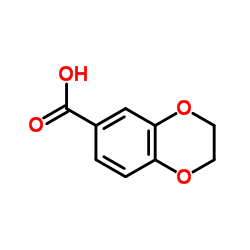 CAS#:4442-54-0
CAS#:4442-54-0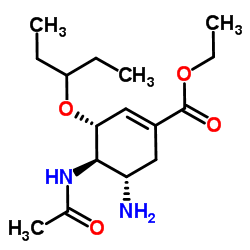 CAS#:196618-13-0
CAS#:196618-13-0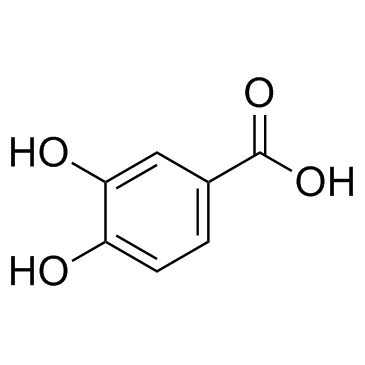 CAS#:99-50-3
CAS#:99-50-3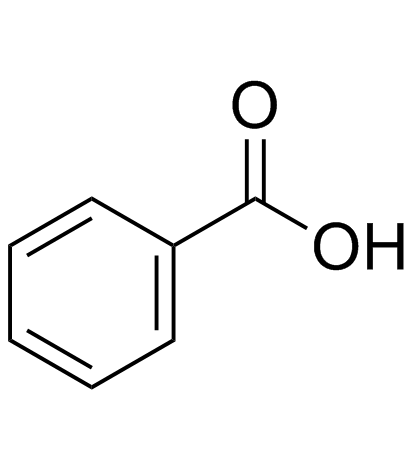 CAS#:65-85-0
CAS#:65-85-0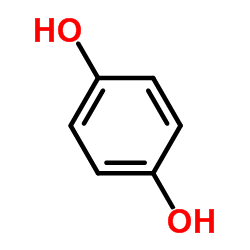 CAS#:123-31-9
CAS#:123-31-9
