Visnagin
Modify Date: 2025-08-25 12:40:12
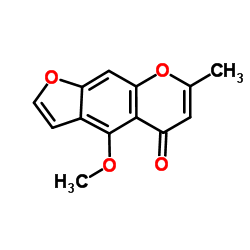
Visnagin structure
|
Common Name | Visnagin | ||
|---|---|---|---|---|
| CAS Number | 82-57-5 | Molecular Weight | 230.216 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 378.2±42.0 °C at 760 mmHg | |
| Molecular Formula | C13H10O4 | Melting Point | 139-142 °C | |
| MSDS | N/A | Flash Point | 182.5±27.9 °C | |
Use of VisnaginVisnagin, an antioxidant furanocoumarin derivative, possess anti-inflammatory and analgesic properties. Visnagin has substantial potential to prevent Cerulein induced acute pancreatitis (AP). Visnagin possess promising vasodilator effects in vascular smooth muscles[1][2]. |
| Name | 4-methoxy-7-methylfuro[3,2-g]chromen-5-one |
|---|---|
| Synonym | More Synonyms |
| Description | Visnagin, an antioxidant furanocoumarin derivative, possess anti-inflammatory and analgesic properties. Visnagin has substantial potential to prevent Cerulein induced acute pancreatitis (AP). Visnagin possess promising vasodilator effects in vascular smooth muscles[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Visnagin (10 µM; for 4, 8, 16, 24 h) induces CYP1A1 transcription in HepG2 cells[1]. Visnagin (10 µM; for 16 h) elevates CYP1B1 gene expression in an aryl hydrocarbon receptor (AHR)-dependent manner, whereas MNF (3’-methoxy-4’-nitroflavone; 20 µM; pre-treated for 1 h) successfully counteracted this induction. Visnagin also enhances PAI-2 transcription in an AHR-dependent manner[1]. |
| In Vivo | Visnagin (10, 30, 60 mg/kg; ip; for 7 days) is effective in reducing plasma amylase and lipase levels and reduces Cerulein (50 μg/kg, six, hourly i.p. injections) induced oxidative stress in male Swiss albino mice (age: 6-8 weeks, weighing 20-25 g)[1]. Visnagin dose dependently decreases the expression of IL-1β, IL-6, TNF-α and IL-17. It attenuates the levels of nuclear p65-NFκB. Visnagin improves the antioxidant defence by improving Nrf2 expression and halts pancreatic inflammation by suppressing NFκB and nitrotyrosine expression in the acinar cells[1]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 378.2±42.0 °C at 760 mmHg |
| Melting Point | 139-142 °C |
| Molecular Formula | C13H10O4 |
| Molecular Weight | 230.216 |
| Flash Point | 182.5±27.9 °C |
| Exact Mass | 230.057907 |
| PSA | 52.58000 |
| LogP | 2.26 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.613 |
| InChIKey | NZVQLVGOZRELTG-UHFFFAOYSA-N |
| SMILES | COc1c2ccoc2cc2oc(C)cc(=O)c12 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Hazard Codes | Xn: Harmful; |
|---|---|
| Risk Phrases | R22 |
| Safety Phrases | S3 |
| RTECS | LV1420000 |
| Precursor 9 | |
|---|---|
| DownStream 7 | |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: Human A3 receptor (Adenosine receptors)
Source: IUPHAR-DB
Target: A3 receptor (Adenosine receptors) [Homo sapiens]
External Id: 21_Human
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
Total 135, Current Page 1 of 14
1
2
3
4
5
| Visnagidin |
| Visnagin |
| 5-methoxy-2-methylfuranobenzopyran-4-one |
| EINECS 201-430-3 |
| Visnacorin |
| Desmethoxykhellin |
| MFCD00005008 |
| Visnagine |
| 5-Methoxy-2-methylfuranochromone |
| 4-Methoxy-7-methyl-5H-furo[3,2-g][1]benzopyran-5-one |
| 4-Methoxy-7-methyl-5H-furo[3,2-g]chromen-5-one |
![4-methoxy-7-methyl-9-(methylthio)-5H-furo[3,2-g]chromen-5-one Structure](https://image.chemsrc.com/caspic/362/122822-92-8.png) CAS#:122822-92-8
CAS#:122822-92-8 CAS#:122822-89-3
CAS#:122822-89-3 CAS#:122822-88-2
CAS#:122822-88-2![7-chloromethyl-4-methoxy-furo[3,2-g]chromen-5-one Structure](https://image.chemsrc.com/caspic/191/70097-62-0.png) CAS#:70097-62-0
CAS#:70097-62-0![7-(Hydroxymethyl)-4-methoxy-5H-furo[3,2-g][1]benzopyran-5-one Structure](https://image.chemsrc.com/caspic/238/478-79-5.png) CAS#:478-79-5
CAS#:478-79-5![4-methoxy-7-(toluene-4-sulfonyloximethyl)-furo[3,2-g]chromen-5-one Structure](https://image.chemsrc.com/caspic/298/855631-87-7.png) CAS#:855631-87-7
CAS#:855631-87-7 CAS#:122822-91-7
CAS#:122822-91-7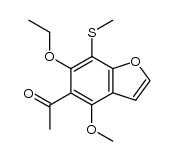 CAS#:122822-90-6
CAS#:122822-90-6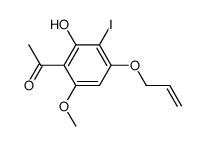 CAS#:74047-33-9
CAS#:74047-33-9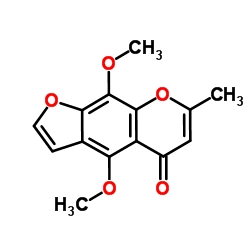 CAS#:82-02-0
CAS#:82-02-0![9-amino-4-hydroxy-7-methylfuro[3,2-g]chromen-5-one structure](https://image.chemsrc.com/caspic/385/16639-42-2.png) CAS#:16639-42-2
CAS#:16639-42-2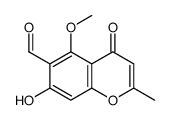 CAS#:7338-51-4
CAS#:7338-51-4 CAS#:88258-42-8
CAS#:88258-42-8 CAS#:484-27-5
CAS#:484-27-5 CAS#:481-71-0
CAS#:481-71-0![[[(4E)-4-(4-methoxy-6-oxo-benzofuran-5-ylidene)-6-methyl-1H-pyrimidin-2-yl]amino]formonitrile structure](https://image.chemsrc.com/caspic/175/52872-86-3.png) CAS#:52872-86-3
CAS#:52872-86-3
