A-740003
Modify Date: 2024-01-02 16:04:09
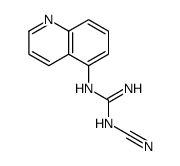
A-740003 structure
|
Common Name | A-740003 | ||
|---|---|---|---|---|
| CAS Number | 861393-28-4 | Molecular Weight | 474.555 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C26H30N6O3 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of A-740003A-740003 is a potent, selective and competitive P2X7 receptor antagonist with IC50 values are 18 and 40 nM for rat and human P2X7 receptors, respectively. |
| Name | N-[1-[(E)-[(cyanoamino)-(quinolin-5-ylamino)methylidene]amino]-2,2-dimethylpropyl]-2-(3,4-dimethoxyphenyl)acetamide |
|---|---|
| Synonym | More Synonyms |
| Description | A-740003 is a potent, selective and competitive P2X7 receptor antagonist with IC50 values are 18 and 40 nM for rat and human P2X7 receptors, respectively. |
|---|---|
| Related Catalog | |
| Target |
IC50: 18 nM (rat P2X7 receptor), 40 nM (human P2X7 receptor) |
| In Vitro | A 438079 or A 740003 (10 μM) significantly blocks the sustained phase of the BzATP-induced response[1]. A-740003 infusion reduces SE-induced TNF-α expression in dentate granule cells. A-740003 infusions increases SE-induced neuronal death[2]. A-740003 and A-438079 show significantly greater potency in blocking P2X7 receptor activation across all species compared with other antagonists. A-740003 and A-438079 show greater activity at rat and human, as compared with mouse P2X7 receptors[3]. A-740003 potently blocks agonist-evoked IL-1β release with (IC50=156 nM) and pore formation (IC50=92 nM) in differentiated human THP-1 cells[4]. |
| In Vivo | Systemic administration of A-740003 produces dose-dependent antinociception in a spinal nerve ligation model (ED50=19 mg/kg i.p.) in the rat. A-740003 also attenuates tactile allodynia in two other models of neuropathic pain, chronic constriction injury of the sciatic nerve and vincristine-induced neuropathy. In addition, A-740003 effectively reduces thermal hyperalgesia observed following intraplantar administration of carrageenan or complete Freund's adjuvant (ED50=38-54 mg/kg i.p.). A-740003 is ineffective in attenuating acute thermal nociception in normal rats and does not alter motor performance at analgesic doses[4]. |
| Animal Admin | The response to acute thermal stimulation is determined using a commercially available paw thermal stimulator. Rats are placed individually in Plexiglas cubicles mounted on a glass surface maintained at 30°C and allowed a 30-min habituation period. A thermal stimulus, in the form of radiant heat emitted from a focused projection bulb, is then applied to the plantar surface of each hind paw. In each test session, each rat is tested in three sequential trials at approximately 5-min intervals. Paw-withdrawal latencies (PWLs) are calculated as the median of the two shortest latencies. An assay cut off is set at 20.5 s. A-740003 is injected i.p. 30 min before testing for acute thermal pain. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Molecular Formula | C26H30N6O3 |
| Molecular Weight | 474.555 |
| Exact Mass | 474.237946 |
| PSA | 120.66000 |
| LogP | 2.77 |
| Index of Refraction | 1.597 |
| Storage condition | 2-8℃ |
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301 |
| Precautionary Statements | P301 + P310 |
| RIDADR | UN 2811 6.1 / PGIII |
|
~% 
A-740003 CAS#:861393-28-4 |
| Literature: Journal of Medicinal Chemistry, , vol. 52, # 10 p. 3366 - 3376 |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
|
Disruption in connexin-based communication is associated with intracellular Ca²⁺ signal alterations in astrocytes from Niemann-Pick type C mice.
PLoS ONE 8 , e71361, (2013) Reduced astrocytic gap junctional communication and enhanced hemichannel activity were recently shown to increase astroglial and neuronal vulnerability to neuroinflammation. Moreover, increasing evide... |
| N-{1-[N''-Cyano-N'-(quinolin-5-yl)carbamimidamido]-2,2-dimethylpropyl}-2-(3,4-dimethoxyphenyl)acetamide |
| N-(1-{(E)-[(Cyanoamino)(5-quinolinylamino)methylene]amino}-2,2-dimethylpropyl)-2-(3,4-dimethoxyphenyl)acetamide |
| cc-516 |
| UNII:532OV6WA05 |
| Benzeneacetamide, N-[1-[[(1E)-(cyanoamino)(5-quinolinylamino)methylene]amino]-2,2-dimethylpropyl]-3,4-dimethoxy- |
| CS-0297 |
| A-740003 |
![N-[1-(1H-Benzotriazol-1-yl)-2,2-dimethylpropyl]-3,4-dimethoxybenzeneacetamide structure](https://www.chemsrc.com/caspic/182/861393-66-0.png)