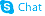O-Desmethylvenlafaxine
O-Desmethylvenlafaxine structure
|
Common Name | O-Desmethylvenlafaxine | ||
|---|---|---|---|---|
| CAS Number | 93413-62-8 | Molecular Weight | 263.375 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 403.8±25.0 °C at 760 mmHg | |
| Molecular Formula | C16H25NO2 | Melting Point | 208-213ºC | |
| MSDS | N/A | Flash Point | 193.2±21.8 °C | |
| Symbol |



GHS02, GHS06, GHS08 |
Signal Word | Danger | |
Use of O-DesmethylvenlafaxineDesvenlafaxine is a serotonin (5-HT) and norepinephrine (NE) reuptake inhibitor with Ki of 40.2 nM and 558.4 nM, respectively. Target: SSRIsDesvenlafaxine is a serotonin-norepinephrine reuptake inhibitor and is the active metabolite of the antidepressant venlafaxine. Similar to venlafaxine, desvenlafaxine inhibits the neuronal uptake of serotonin and norepinephrine. Desvenlafaxine shows weak binding affinity (62% inhibition at 100 μM) at the human dopamine (DA) transporter. Desvenlafaxine inhibits [3H]5-HT or [3H]NE uptake for the hSERT or hNET with IC50 of 47.3 and 531.3 nM, respectively. Desvenlafaxine rapidly penetrates the male rat brain and hypothalamus. Desvenlafaxine significantly increases extracellular NE levels compared with baseline in the male rat hypothalamus but had no effect on DA levels using microdialysis [1]. Desvenlafaxine has the potential to inhibit CYP2D6, which could result in increased concentrations of drugs metabolized through this pathway. Induction of CYP3A4 is also possible with desvenlafaxine, which could impact the metabolism of drugs metabolized via this enzyme. Desvenlafaxine exhibits a linear and dose-proportional pharmacokinetic single-dose profile in a dose range from 100 to 600 mg/day. The absolute bioavailability of the oral formulation is 80.5% [2]. |
| Name | O-desmethylvenlafaxine |
|---|---|
| Synonym | More Synonyms |
| Description | Desvenlafaxine is a serotonin (5-HT) and norepinephrine (NE) reuptake inhibitor with Ki of 40.2 nM and 558.4 nM, respectively. Target: SSRIsDesvenlafaxine is a serotonin-norepinephrine reuptake inhibitor and is the active metabolite of the antidepressant venlafaxine. Similar to venlafaxine, desvenlafaxine inhibits the neuronal uptake of serotonin and norepinephrine. Desvenlafaxine shows weak binding affinity (62% inhibition at 100 μM) at the human dopamine (DA) transporter. Desvenlafaxine inhibits [3H]5-HT or [3H]NE uptake for the hSERT or hNET with IC50 of 47.3 and 531.3 nM, respectively. Desvenlafaxine rapidly penetrates the male rat brain and hypothalamus. Desvenlafaxine significantly increases extracellular NE levels compared with baseline in the male rat hypothalamus but had no effect on DA levels using microdialysis [1]. Desvenlafaxine has the potential to inhibit CYP2D6, which could result in increased concentrations of drugs metabolized through this pathway. Induction of CYP3A4 is also possible with desvenlafaxine, which could impact the metabolism of drugs metabolized via this enzyme. Desvenlafaxine exhibits a linear and dose-proportional pharmacokinetic single-dose profile in a dose range from 100 to 600 mg/day. The absolute bioavailability of the oral formulation is 80.5% [2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 403.8±25.0 °C at 760 mmHg |
| Melting Point | 208-213ºC |
| Molecular Formula | C16H25NO2 |
| Molecular Weight | 263.375 |
| Flash Point | 193.2±21.8 °C |
| Exact Mass | 263.188538 |
| PSA | 43.70000 |
| LogP | 2.26 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.573 |
| Storage condition | -20°C Freezer |
| Symbol |



GHS02, GHS06, GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H225-H301 + H311 + H331-H370 |
| Precautionary Statements | P210-P260-P280-P301 + P310-P311 |
| Hazard Codes | F,T,Xi |
| Risk Phrases | 11-23/24/25-39/23/24/25-36 |
| Safety Phrases | 7-16-36/37-45-26 |
| RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution |
| RTECS | GV8872620 |
| HS Code | 2922509090 |
| Precursor 9 | |
|---|---|
| DownStream 3 | |
| HS Code | 2922509090 |
|---|---|
| Summary | 2922509090. other amino-alcohol-phenols, amino-acid-phenols and other amino-compounds with oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Evaluation of the matrix effect of different sample matrices for 33 pharmaceuticals by post-column infusion.
J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 1000 , 84-94, (2015) Matrix effects that occur during quantitative measurement by liquid chromatography mass spectrometry specifically when using electrospray ionization are a widely recognized phenomenon. Sample matrix c... |
|
|
Hollow-fiber liquid-phase microextraction and chiral LC-MS/MS analysis of venlafaxine and its metabolites in plasma.
Bioanalysis 5(6) , 721-30, (2013) An enantioselective analytical method was developed and validated for determination of venlafaxine and its metabolites O-desmethylvenlafaxine and N-desmethylvenlafaxine in plasma samples. The method e... |
|
|
A poor metabolizer of both CYP2C19 and CYP2D6 identified by mechanistic pharmacokinetic simulation in a fatal drug poisoning case involving venlafaxine.
Forensic Sci. Int. 226(1-3) , e26-31, (2013) We present a fatal drug poisoning case involving venlafaxine (VEN). The deceased took his medication regularly (including 150 mg VEN twice daily), and nothing in the case or autopsy findings pointed t... |
| Phenol, 4-[2-(dimethylamino)-1-(1-hydroxycyclohexyl)ethyl]- |
| desvenlafaxine |
| O-Desmethylvenlafaxine |
| L6TJ AYR DQ&1N1&1 AQ |
| 4-[2-(Dimethylamino)-1-(1-hydroxycyclohexyl)ethyl]phenol |
| 4-(2-(Dimethylamino)-1-(1-hydroxycyclohexyl)ethyl)phenol |
![1-[1-(4-Benzyloxyphenyl)-2-(dimethylamino)ethyl]cyclohexanol Structure](https://www.chemsrc.com/caspic/422/93413-61-7.png) CAS#:93413-61-7
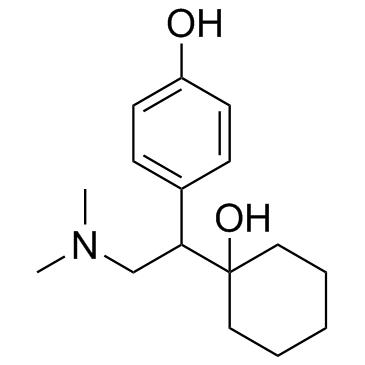
CAS#:93413-61-7 CAS#:93413-69-5
CAS#:93413-69-5 CAS#:1000377-24-1
CAS#:1000377-24-1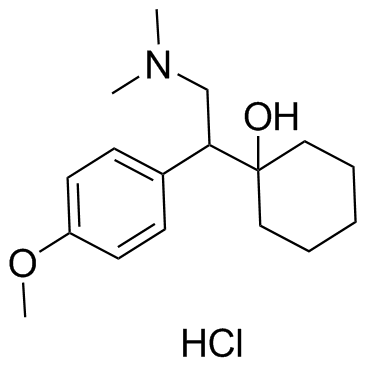 CAS#:99300-78-4
CAS#:99300-78-4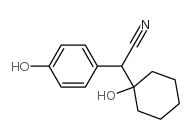 CAS#:918344-20-4
CAS#:918344-20-4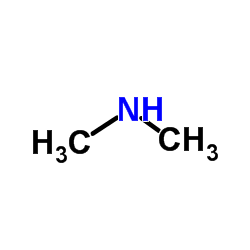 CAS#:124-40-3
CAS#:124-40-3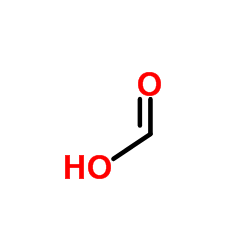 CAS#:64-18-6
CAS#:64-18-6![5-(4-Methoxyphenyl)-3-methyl-1-oxa-3-azaspiro[5.5]undecane hydroc hloride Structure](https://www.chemsrc.com/caspic/429/93413-56-0.png) CAS#:93413-56-0
CAS#:93413-56-0 CAS#:149289-29-2
CAS#:149289-29-2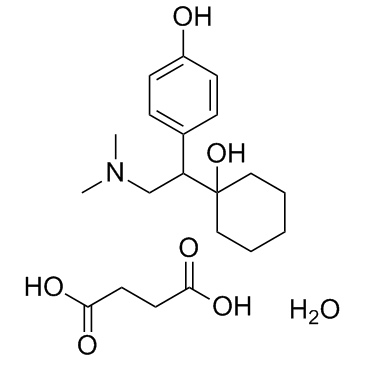 CAS#:386750-22-7
CAS#:386750-22-7 CAS#:300827-87-6
CAS#:300827-87-6 CAS#:448904-47-0
CAS#:448904-47-0