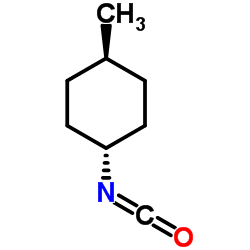
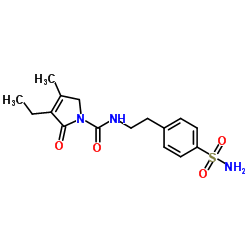
Glimepiride

Glimepiride structure
|
Common Name | Glimepiride | ||
|---|---|---|---|---|
| CAS Number | 93479-97-1 | Molecular Weight | 490.616 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 677.0±65.0 °C at 760 mmHg | |
| Molecular Formula | C24H34N4O5S | Melting Point | 212.2-214.5 °C | |
| MSDS | Chinese USA | Flash Point | 363.2±34.3 °C | |
Use of GlimepirideGlimepiride(Amaryl) is a medium-to-long acting sulfonylurea anti-diabetic compound with an ED50 of 182 μg/kg.Target: DPP4Glimepiride (Hoe 490) is a new sulfonylurea. After oral administration of Hoe 490 to rabbits, blood glucose was lowered 3.5 times more than after glibenclamide (HB 419) and after intravenous administration, 2.5 times more [1]. glimepiride decreased extracellular Aβ40 and Aβ42 levels. glimepiride may serve as a promising drug for the treatment of AD associated with diabetes [2]. glimepiride was generally associated with lower risk of hypoglycemia and less weight gain compared to other sulfonylureas. Glimepiride use may be safer in patients with cardiovascular disease because of its lack of detrimental effects on ischemic preconditioning [3].Clinical indications: Non-insulin dependent diabetesToxicity: Severe hypoglycemic reactions with coma; seizure or other neurological impairment. |
| Name | glimepiride |
|---|---|
| Synonym | More Synonyms |
| Description | Glimepiride(Amaryl) is a medium-to-long acting sulfonylurea anti-diabetic compound with an ED50 of 182 μg/kg.Target: DPP4Glimepiride (Hoe 490) is a new sulfonylurea. After oral administration of Hoe 490 to rabbits, blood glucose was lowered 3.5 times more than after glibenclamide (HB 419) and after intravenous administration, 2.5 times more [1]. glimepiride decreased extracellular Aβ40 and Aβ42 levels. glimepiride may serve as a promising drug for the treatment of AD associated with diabetes [2]. glimepiride was generally associated with lower risk of hypoglycemia and less weight gain compared to other sulfonylureas. Glimepiride use may be safer in patients with cardiovascular disease because of its lack of detrimental effects on ischemic preconditioning [3].Clinical indications: Non-insulin dependent diabetesToxicity: Severe hypoglycemic reactions with coma; seizure or other neurological impairment. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 677.0±65.0 °C at 760 mmHg |
| Melting Point | 212.2-214.5 °C |
| Molecular Formula | C24H34N4O5S |
| Molecular Weight | 490.616 |
| Flash Point | 363.2±34.3 °C |
| Exact Mass | 490.224976 |
| PSA | 133.06000 |
| LogP | 4.17 |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.628 |
| Storage condition | Room temp |
| Water Solubility | DMSO: >10 mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xn,Xi |
| Risk Phrases | R21 |
| Safety Phrases | S25-S26-S36/37-S53 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UX9363950 |
| HS Code | 2935009090 |
|
~86% 
Glimepiride CAS#:93479-97-1 |
| Literature: WO2006/103690 A1, ; Page/Page column 7,16 ; |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
| HS Code | 2935009090 |
|---|---|
| Summary | 2935009090 other sulphonamides VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:35.0% |
|
Drug-drug interaction of losartan and glimepiride metabolism by recombinant microsome CYP2C9*1, 2C9*3, 2C9*13, and 2C9*16 in vitro.
Int. J. Clin. Pharmacol. Ther. 52(9) , 732-8, (2014) Co-administration of anti-hypertension and anti-diabetic drugs is common in clinical settings.In this study, we characterized the drug-drug interactions of losartan (LOS) and glimepiride (GLP) using r... |
|
|
Role of wetland organic matters as photosensitizer for degradation of micropollutants and metabolites.
J. Hazard. Mater. 276 , 1-9, (2014) Overall photodegradation of pharmaceuticals, personal care products (PPCPs) and pharmaceutical metabolites were investigated in order to evaluate their photochemical fate in aquatic environments in va... |
|
|
Analysis of multi-site drug-protein interactions by high-performance affinity chromatography: Binding by glimepiride to normal or glycated human serum albumin.
J. Chromatogr. A. 1408 , 133-44, (2015) High-performance affinity chromatography (HPAC) was used in a variety of formats to examine multi-site interactions between glimepiride, a third-generation sulfonylurea drug, and normal or in vitro gl... |
| Glimpiride |
| Glimepiride |
| Grimepride |
| amary |
| AMARYL |
| GliMepiride COS |
| CLIMEPIRIDE |
| MFCD00878417 |
| glimepirid |
| GliMperide |
| hoe490 |