4,6-二氯-5-氨基嘧啶
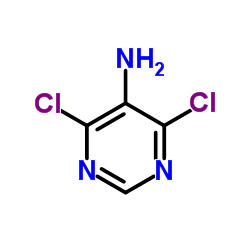
4,6-二氯-5-氨基嘧啶结构式

|
常用名 | 4,6-二氯-5-氨基嘧啶 | 英文名 | 4,6-Dichloro-5-pyrimidinamine |
|---|---|---|---|---|
| CAS号 | 5413-85-4 | 分子量 | 163.99 | |
| 密度 | 1.6±0.1 g/cm3 | 沸点 | 267.1±35.0 °C at 760 mmHg | |
| 分子式 | C4H3Cl2N3 | 熔点 | 145-148 °C(lit.) | |
| MSDS | 美版 | 闪点 | 115.3±25.9 °C | |
| 符号 |

GHS07 |
信号词 | Warning |
|
6-(Alkylamino)-9-benzyl-9H-purines. A new class of anticonvulsant agents.
J. Med. Chem. 31(3) , 606-12, (1988) Several 9-alkyl-6-substituted-purines were synthesized and tested for anticonvulsant activity against maximal electroshock-induced seizures (MES) in rats. Most compounds were prepared in three steps from 5-amino-4,6-dichloropyrimidine or in two steps via alky... |
|
|
An efficient and regiospecific strategy to N7-substituted purines and its application to a library of trisubstituted purines.
J. Comb. Chem. 8(3) , 410-6, (2006) A regiospecific strategy for the preparation of N(7)-substituted purines in an efficient manner was devised. This approach to 6,7,8-trisubstituted purines relies on the cyclization reactions of suitably substituted pyrimidines (1) with either a carboxylic aci... |
|
|
Adenosine deaminase inhibitors. Synthesis and biological evaluation of C1' and nor-C1' derivatives of (+)-erythro-9-(2(S)-hydroxy-3(R)-nonyl)adenine.
J. Med. Chem. 35(22) , 4180-4, (1992) The synthesis of various chiral derivatives of (+)-erythro-9-(2-hydroxy-3-nonyl)adenine, (+)-EHNA, from (2S,3R)-3-amino-1,2-O-isopropylidene-1,2-nonanediol by condensation with 5-amino-4,6-dichloropyrimidine is described. The compounds synthesized were C1'- a... |
|
|
Synthesis of oxepane ring containing monocyclic, conformationally restricted bicyclic and spirocyclic nucleosides from D-glucose: a cycloaddition approach.
J. Org. Chem. 72(19) , 7427-30, (2007) Carbohydrate-derived substrates having (i) C-5 nitrone and C-3-O-allyl, (ii) C-4 vinyl and a C-3-O-tethered nitrone, and (iii) C-5 nitrone and C-4-allyloxymethyl generated tetracyclic isoxazolidinooxepane/-pyran ring systems upon intramolecular nitrone cycloa... |
|
|
Sequential ring-closing metathesis and nitrone cycloaddition on glucose-derived substrates: a divergent approach to analogues of spiroannulated carbanucleosides and conformationally locked nucleosides.
J. Org. Chem. 71(16) , 5980-92, (2006) The carbohydrate-derived substrate 3-C-allyl-1,2:5,6-di-O-isopropylidene-alpha-D-allofuranose was judiciously manipulated for preparing suitable synthons, which could be converted to a variety of isoxazolidino-spirocycles and -tricycles through the applicatio... |
|
|
Tetrahedron Lett. 48 , 1489, (2007)
|