几夫碱
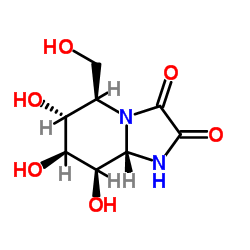
几夫碱结构式

|
常用名 | 几夫碱 | 英文名 | Kifunensine |
|---|---|---|---|---|
| CAS号 | 109944-15-2 | 分子量 | 232.191 | |
| 密度 | 1.9±0.1 g/cm3 | 沸点 | N/A | |
| 分子式 | C8H12N2O6 | 熔点 | >280℃ | |
| MSDS | 中文版 美版 | 闪点 | N/A |
|
A context-independent N-glycan signal targets the misfolded extracellular domain of Arabidopsis STRUBBELIG to endoplasmic-reticulum-associated degradation.
Biochem. J. 464(3) , 401-11, (2014) N-glycosylation of proteins plays an important role in the determination of the fate of newly synthesized glycoproteins in the endoplasmic reticulum (ER). Specific oligosaccharide structures recruit molecular chaperones that promote folding or mannose-binding... |
|
|
Synthesis of kifunensine thioanalogs and their inhibitory activities against HIV-RT and α-mannosidase.
Carbohydr. Res. 365 , 1-8, (2013) An efficient and practical synthesis of kifunensine thioanalogs 1a-c was reported. The bicyclic azasugars fused thiazolidin-4-one 4a-c as key intermediates were first synthesized in good yields of 74-80% via one-pot tandem Staudinger/aza-Wittig/cyclization by... |
|
|
Evolution of cross-neutralizing antibody specificities to the CD4-BS and the carbohydrate cloak of the HIV Env in an HIV-1-infected subject.
PLoS ONE 7(11) , e49610, (2012) Broadly neutralizing antibodies are considered an important part of a successful HIV vaccine. A better understanding of the factors underlying their development during infection and of the epitopes they target is needed to elicit similar antibody responses by... |
|
|
The unfolded protein response transducer ATF6 represents a novel transmembrane-type endoplasmic reticulum-associated degradation substrate requiring both mannose trimming and SEL1L protein.
J. Biol. Chem. 288(44) , 31517-27, (2013) Proteins misfolded in the endoplasmic reticulum (ER) are cleared by the ubiquitin-dependent proteasome system in the cytosol, a series of events collectively termed ER-associated degradation (ERAD). It was previously shown that SEL1L, a partner protein of the... |
|
|
Mannosidase I inhibition rescues the human alpha-sarcoglycan R77C recurrent mutation.
Hum. Mutat. 17 , 1214-21, (2008) Limb girdle muscular dystrophy type 2D (LGMD2D, OMIM600119) is a genetic progressive myopathy that is caused by mutations in the human alpha-sarcoglycan gene (SGCA). Here, we have introduced in mice the most prevalent LGMD2D mutation, R77C. It should be noted... |
|
|
Lack of the Golgi phosphate transporter PHT4;6 causes strong developmental defects, constitutively activated disease resistance mechanisms and altered intracellular phosphate compartmentation in Arabidopsis.
Plant J. 72(5) , 732-44, (2012) The Golgi-located phosphate exporter PHT4;6 has been described as involved in salt tolerance but further analysis on the physiological impact of PHT4;6 remained elusive. Here we show that PHT4;6-GFP is targeted to the trans-Golgi compartment and that loss of ... |
|
|
Epitope specificity of cross-clade neutralizing sera from Chinese HIV-1-positive individuals.
Scand. J. Immunol. 78(4) , 357-70, (2013) Induction of broadly neutralizing antibody is considered important for an effective HIV-1 vaccine. Identification and characterization of broadly neutralizing antibodies in HIV-1-infected patients will facilitate our understanding of the immune correlates to ... |
|
|
TGF-β sensitivity is determined by N-linked glycosylation of the type II TGF-β receptor.
Biochem. J. 445(3) , 403-11, (2012) N-linked glycosylation is a critical determinant of protein structure and function, regulating processes such as protein folding, stability and localization, ligand-receptor binding and intracellular signalling. TβRII [type II TGF-β (transforming growth facto... |
|
|
MHCII glycosylation modulates Bacteroides fragilis carbohydrate antigen presentation.
J. Exp. Med. 208(5) , 1041-53, (2011) N-linked glycans are thought to protect class II major histocompatibility complex (MHC) molecules (MHCII) from proteolytic cleavage and assist in arranging proteins within the immune synapse, but were not thought to directly participate in antigen presentatio... |
|
|
Inhibition of endoplasmic reticulum-associated degradation rescues native folding in loss of function protein misfolding diseases.
J. Biol. Chem. 286(50) , 43454-64, (2011) Lysosomal storage disorders are often caused by mutations that destabilize native folding and impair trafficking of secretory proteins. We demonstrate that endoplasmic reticulum (ER)-associated degradation (ERAD) prevents native folding of mutated lysosomal e... |