L-798106
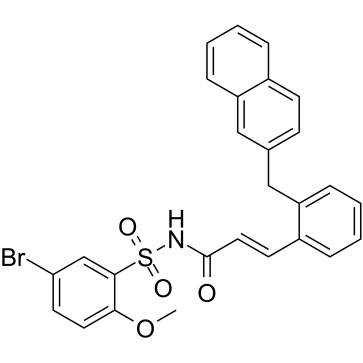
L-798106结构式

|
常用名 | L-798106 | 英文名 | L-798106 |
|---|---|---|---|---|
| CAS号 | 244101-02-8 | 分子量 | 536.43700 | |
| 密度 | 1.425±0.06 g/cm3(Predicted) | 沸点 | N/A | |
| 分子式 | C27H22BrNO4S | 熔点 | N/A | |
| MSDS | 中文版 美版 | 闪点 | N/A |
L-798106用途L-798106 是强效且高度选择性的前列腺素类 EP3 受体拮抗剂 (Ki=0.3 nM),它在 EP4,EP1 和 EP2 受体上也具有微摩尔活性,针对 EP4,EP1,EP2 的 Ki 值分别为 916 nM,>5000 nM 和 >5000 nM。 |
| 中文名 | (2E)-N-[(5-溴-2-甲氧基苯基)磺酰基]-3-[2-(2-萘甲基)苯基]-2-丙烯酰胺 |
|---|---|
| 英文名 | N-(5-bromo-2-methoxyphenyl)sulfonyl-3-[2-(naphthalen-2-ylmethyl)phenyl]prop-2-enamide |
| 英文别名 | 更多 |
| 描述 | L-798106 是强效且高度选择性的前列腺素类 EP3 受体拮抗剂 (Ki=0.3 nM),它在 EP4,EP1 和 EP2 受体上也具有微摩尔活性,针对 EP4,EP1,EP2 的 Ki 值分别为 916 nM,>5000 nM 和 >5000 nM。 |
|---|---|
| 相关类别 | |
| 靶点实验 |
Ki values: 0.3 nM (EP3 receptor); 916 nM (EP4 receptor); > 5000 nM (EP1/2 receptor)[1] |
| 参考文献 |
| 密度 | 1.425±0.06 g/cm3(Predicted) |
|---|---|
| 分子式 | C27H22BrNO4S |
| 分子量 | 536.43700 |
| 精确质量 | 535.04500 |
| PSA | 80.85000 |
| LogP | 7.19170 |
| InChIKey | ODTKFNUPVBULRJ-NTCAYCPXSA-N |
| SMILES | COc1ccc(Br)cc1S(=O)(=O)NC(=O)C=Cc1ccccc1Cc1ccc2ccccc2c1 |
| 储存条件 | 2-8°C |
| 危害声明 | H413 |
|---|---|
| 危险品运输编码 | NONH for all modes of transport |
|
Angiogenesis in the primate ovulatory follicle is stimulated by luteinizing hormone via prostaglandin E2.
Biol. Reprod. 92(1) , 15, (2015) Rapid angiogenesis occurs as the ovulatory follicle is transformed into the corpus luteum. To determine if luteinizing hormone (LH)-stimulated prostaglandin E2 (PGE2) regulates angiogenesis in the ovu... |
|
|
Mechanisms for proteinase-activated receptor 1-triggered prostaglandin E2 generation in mouse osteoblastic MC3T3-E1 cells.
Biol. Chem. 396(2) , 153-62, (2015) We analyzed signaling mechanisms for prostaglandin E2 (PGE2) production following activation of proteinase-activated receptor-1 (PAR1), a thrombin receptor, in preosteoblastic MC3T3-E1 cells. PAR1 sti... |
|
|
6-Substituted 9-fluoroquino[3,2-b]benzo[1,4]thiazines display strong antiproliferative and antitumor properties.
Eur. J. Med. Chem. 89 , 411-20, (2014) 6-Substituted 9-fluoroquino[3,2-b]benzo[1,4]thiazines - a new type of tetracyclic azaphenothiazines-were obtained from of 6H-9-fluoroquinobenzothiazine by the introduction of appropriate substituents ... |
| (2E)-N-[(5-bromo-2-methoxyphenyl)sulfonyl]-3-[2-(2-naphthalenylmethyl)phenyl]-2-propenamide |
| Zinterol hydrochloride |







